Overcoming Tumor Vessel Heterogeneity: Strategies for Effective Drug Delivery and Therapeutic Targeting
This article addresses the critical challenge of tumor vasculature heterogeneity in oncology research and drug development.
Overcoming Tumor Vessel Heterogeneity: Strategies for Effective Drug Delivery and Therapeutic Targeting
Abstract
This article addresses the critical challenge of tumor vasculature heterogeneity in oncology research and drug development. We first explore the biological foundations of abnormal tumor vessel structure and function, detailing how spatial and temporal heterogeneity arises and impedes treatment. We then review current and emerging methodological approaches, from vascular normalization and permeability enhancement to novel carrier systems, designed to compensate for this variability. Practical troubleshooting sections address common experimental pitfalls and optimization strategies for in vivo models and imaging. Finally, we provide a comparative analysis of validation techniques and clinical translation frameworks. This comprehensive guide synthesizes cross-disciplinary knowledge to equip researchers and drug developers with strategies to overcome vascular barriers and improve therapeutic outcomes.
Decoding the Puzzle: Understanding the Biology of Heterogeneous Tumor Vasculature
Technical Support Center: Troubleshooting & FAQs
Q1: In our perfusion experiment, we see highly variable dye uptake across the tumor. Does this indicate a problem with our injection technique or confirm functional heterogeneity? A: Variable uptake is a hallmark of functional heterogeneity. First, rule out technical issues:
- Check Injection Consistency: Ensure a stable, slow bolus via tail-vein catheter over 5-7 seconds. Rapid manual injection causes streaming artifacts. Use a syringe pump for reproducibility.
- Control for Cardiac Output: Animal heart rate and anesthesia depth (e.g., 1.5-2% isoflurane) dramatically affect perfusion. Monitor vitals.
- Quantify the Heterogeneity: Calculate the Coefficient of Variation (CV = Standard Deviation / Mean) of fluorescence intensity across multiple tumor regions. A CV > 30% in controlled conditions strongly indicates biological heterogeneity, not technical error.
Q2: Our immunohistochemistry (IHC) for endothelial markers (CD31) shows uneven staining, with some vessels appearing fragmented. Is this a fixation artifact or structural abnormality? A: This is a common challenge. To differentiate:
- Optimize Fixation: Over-fixation (>24h in formalin) can mask epitopes. For CD31, limit fixation to 18-24h at 4°C. Perform antigen retrieval using pH 9.0 EDTA buffer with a pressure cooker for 10 minutes.
- Use a Complementary Stain: Co-stain for a basement membrane component (Collagen IV). If Collagen IV forms a continuous sleeve where CD31 is fragmented, it indicates a structurally immature or regressing vessel—a key feature of structural heterogeneity. If both are discontinuous, consider artifact.
- Include a Positive Control: Normal tissue (e.g., muscle) should show continuous, uniform staining.
Q3: When measuring vessel permeability using fluorescent dextrans, background signal is too high. How can we improve the signal-to-noise ratio? A: High background is often due to slow clearance or extravascular trapping.
- Protocol Adjustment: Use a circulation time of 3-5 minutes for 70 kDa Texas Red-dextran. Perfuse with 10-20 mL of PBS immediately after to flush non-bound tracer from the vasculature.
- Switch Tracer Size: If studying enhanced permeability and retention (EPR), use a larger dextran (155 kDa or 2000 kDa). Smaller dextrans (4-40 kDa) diffuse rapidly, increasing background.
- Data Correction: Subtract the mean fluorescence intensity of a non-vascular, non-necrotic region from your vessel-associated measurements.
Q4: Our flow cytometry data on dissociated tumor endothelial cells (CD45-/CD31+) shows a wide spread in expression levels. How do we gate this population correctly? A: Heterogeneity in marker expression is expected. Use a systematic gating strategy:
- Live/Dead Exclusion: Use Zombie NIR or DAPI.
- Lineage Exclusion: Gate out CD45+ (leukocytes) and Ter119+ (erythrocytes).
- Endothelial Enrichment: Plot CD31 vs. CD102 (ICAM-2). True tumor endothelial cells (TECs) will be CD31+/CD102+. Use fluorescence-minus-one (FMO) controls to set gates for low-expressors.
- Sub-population Analysis: Further characterize by staining for markers of heterogeneity like VEGFR2 (phosho-VEGFR2 for activity), CD105 (Endoglin for angiogenesis), or CXCR4.
Table 1: Key Quantitative Metrics for Assessing Vasculature Heterogeneity
| Category | Specific Metric | Typical Measurement Technique | Normal Tissue Range (Approx.) | Heterogeneous Tumor Range (Approx.) | Interpretation |
|---|---|---|---|---|---|
| Structural | Vessel Density | CD31 IHC, confocal microscopy | 200-400 vessels/mm² | 100-600 vessels/mm² | High spatial variability. |
| Structural | Pericyte Coverage Index | α-SMA+/CD31+ area ratio (IHC) | 70-90% | 10-70% | Low index indicates immaturity. |
| Structural | Vessel Diameter Distribution | Histology, micro-CT | Tight distribution (~5-10µm) | Wide distribution (5-50µm) | Presence of chaotic, dilated vessels. |
| Functional | Perfusion Efficiency | Lectin or fluorescent dye uptake | >95% vessels perfused | 20-60% vessels perfused | High fraction of non-functional vessels. |
| Functional | Permeability Coefficient (P) | Evans Blue, 70 kDa dextran extravasation | P < 1.0 x 10⁻⁷ cm/s | P = 1.0-50.0 x 10⁻⁷ cm/s | Elevated and variable permeability. |
| Functional | Hypoxic Fraction | Pimonidazole adducts IHC | <10% tissue area | 20-60% tissue area | Correlates with poor perfusion. |
Detailed Experimental Protocols
Protocol 1: Multiparametric In Vivo Perfusion and Permeability Assay
- Objective: Simultaneously quantify functional perfusion and permeability heterogeneity.
- Materials: See "Research Reagent Solutions" below.
- Method:
- Tracer Cocktail Injection: Via tail vein catheter, inject a mixture of FITC-Lectin (50 µg in 100 µL PBS, labels perfused vasculature) and Texas Red-Dextran 155 kDa (25 mg/mL, 100 µL, assesses permeability).
- Circulation: Allow lectin to circulate for 3 minutes. Dextran circulates for 20 minutes.
- Vascular Washout: Euthanize animal. Perfuse transcardially with 20 mL ice-cold PBS at 120 mmHg to flush intravascular dextran.
- Tumor Harvest & Processing: Excise tumor, snap-freeze in O.C.T. Cryosection (30 µm).
- Imaging & Analysis: Acquire z-stacks via confocal microscope. Use ImageJ to:
- Calculate % Perfused Vessels: (FITC+ CD31+ area / Total CD31+ area) x 100.
- Calculate Permeability Index: (Texas Red extravascular intensity / Intravascular Texas Red intensity) in lectin-positive vessels.
Protocol 2: Spatial Mapping of Vascular Phenotypes via Multiplex IHC
- Objective: Correlate structural features (maturity) with functional status in the same tissue section.
- Method:
- Tumor Preparation: From Protocol 1, fix some tumor sections in 4% PFA for IHC.
- Staining Panel: Perform sequential IHC/IF for:
- Round 1: CD31 (Endothelial), α-SMA (Pericytes), DAPI.
- Round 2: After mild stripping, stain for pimonidazole (Hypoxia) and Collagen IV (Basement Membrane).
- Image Registration: Use software (e.g., HALO, QuPath) to align images from both rounds.
- Spatial Analysis: Classify vessels into phenotypes:
- Mature & Perfused: CD31+, α-SMA High, Collagen IV+, Pimo-.
- Immature & Hypoxic: CD31+, α-SMA Low, Collagen IV-/low, Pimo+.
Signaling Pathways in Vascular Heterogeneity
Title: Key Signaling Pathways Driving Vascular Heterogeneity
Title: Integrated Workflow for Assessing Vascular Heterogeneity
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Tumor Vasculature Heterogeneity Studies
| Reagent | Category | Specific Example | Function in Experiment |
|---|---|---|---|
| Fluorescent Lectin | Perfusion Tracer | Lycopersicon esculentum (Tomato) Lectin, FITC conjugate | Binds selectively to glycosylated surfaces of perfused endothelial cells. |
| Size-Fractionated Dextrans | Permeability Tracer | Texas Red-Dextran, 70 kDa and 155 kDa | Measures vessel leakiness; different sizes probe different pore sizes. |
| Hypoxia Marker | Chemical Probe | Pimonidazole HCl | Forms protein adducts in cells with pO₂ < 10 mmHg, detectable by IHC. |
| Endothelial Marker | Antibody (IHC/Flow) | Anti-CD31 (PECAM-1) | Primary marker for identifying blood vessel endothelial cells. |
| Pericyte Marker | Antibody (IHC) | Anti-Alpha Smooth Muscle Actin (α-SMA) | Identifies vascular smooth muscle cells and pericytes for maturity assessment. |
| Basement Membrane Marker | Antibody (IHC) | Anti-Collagen IV | Labels the basement membrane, indicating vessel stability and maturity. |
| Viability Dye | Flow Cytometry | Zombie NIR Fixable Viability Kit | Distinguishes live from dead cells during endothelial cell isolation for FACS. |
Technical Support Center
Troubleshooting Guides & FAQs
FAQ Category 1: Hypoxia Chamber & Induction Experiments
Q1: Our hypoxic cell cultures (e.g., 1% O₂) show inconsistent HIF-1α stabilization across replicates. What are the primary causes and solutions? A: Inconsistent HIF-1α stabilization is commonly due to:
- Chamber Seal Failure: Ensure the airlock is properly sealed and the gas regulator is calibrated. Perform weekly checks with an independent O₂ sensor probe.
- Media Overfill: Excessive media volume slows O₂ diffusion. Maintain a media depth of ≤5mm.
- Cell Confluence Variability: Seed cells at a uniform density (±5% variance). High confluence creates localized nutrient gradients.
- Protocol: Pre-equilibrate media in the hypoxia chamber for 4 hours before adding to cells. Use a chemical hypoxia inducer (e.g., CoCl₂ at 150µM) as a positive control in parallel.
Q2: How do we accurately quantify and map a VEGF gradient in a 3D tumor spheroid or co-culture model? A: Direct measurement requires specialized techniques. Use this sequential protocol: 1. Sample Fixation: Fix spheroids in 4% PFA for 45 minutes at room temperature under hypoxic conditions. 2. Microdissection & ELISA: Cryosection spheroid into concentric rings (e.g., outer, middle, core) using a cryostat. Pool corresponding rings from 10-15 spheroids and perform a high-sensitivity VEGF ELISA. 3. Data Normalization: Normalize VEGF concentration to total protein content per ring (Bradford assay).
Q3: Our fibroblast (CAF) co-culture experiments are yielding high background VEGF, masking tumor cell-specific secretion. How can we isolate the contributions? A: Use a transwell system with genetic tagging. * Protocol: Seed fluorescently tagged (e.g., GFP) tumor cells in the lower chamber. Seed CAFs in the transwell insert. After hypoxic incubation, collect media separately from upper and lower chambers. Analyze VEGF via ELISA and attribute source via cell tag. Use species-specific VEGF antibodies if co-culturing human and mouse cells.
FAQ Category 2: VEGF Signaling & Inhibition
Q4: Despite using a VEGFR-2 inhibitor (e.g., SU5416), we still observe phosphorylated ERK in tumor cells. What are possible resistance mechanisms? A: This indicates compensatory signaling bypassing VEGFR-2. * Checkpoints: 1. Alternative VEGF Receptors: Probe for p-VEGFR-1 and neuropilin-1 (NRP1) activity. 2. Stromal Feedback: CAFs may secrete alternative ligands (e.g., FGF2, PDGF). Perform conditioned media transfer experiments. 3. Off-target Akt/mTOR activation: Analyze p-Akt and p-S6K levels.
Q5: What is the optimal method for validating VEGF gradient function in a migration assay? A: Implement a under-agarose assay with a VEGF trap control. * Protocol: 1. Prepare a 2% agarose gel in serum-free media in a 6-well plate. 2. Punch three wells: center (for cells), source (for VEGF, e.g., 50ng/mL), and control (for VEGF + 10µg/mL VEGF Trap). 3. Seed GFP-labeled cells (e.g., endothelial or tumor) in the center well. 4. Image migration directionality and distance over 24h. Directional migration toward the VEGF source that is abrogated in the Trap well confirms gradient functionality.
Table 1: Hypoxia-Induced VEGF Secretion Across Cell Types
| Cell Type | Normoxic VEGF (pg/mL/10⁶ cells/24h) | Hypoxic (1% O₂) VEGF (pg/mL/10⁶ cells/24h) | Fold Increase | Primary Receptor Expressed |
|---|---|---|---|---|
| Human Umbilical Vein ECs (HUVECs) | 150 ± 25 | 450 ± 75 | 3.0 | VEGFR-2 |
| Glioblastoma (U87-MG) | 1200 ± 150 | 4800 ± 350 | 4.0 | VEGFR-1/NRP1 |
| Carcinoma-Associated Fibroblasts | 850 ± 100 | 2500 ± 300 | 2.9 | VEGFR-2/VEGFR-3 |
| Renal Carcinoma (786-O) | 3000 ± 400 | 3500 ± 450 | 1.2 | VEGFR-2 |
Table 2: Efficacy of VEGF Pathway Inhibitors in Heterogeneous Co-culture
| Inhibitor (10µM) | Target | Reduction in HUVEC Tubulogenesis (%) | Reduction in CAF-Mediated Invasion (%) | Impact on Spheroid Core Viability (%) |
|---|---|---|---|---|
| SU5416 | VEGFR-2 | 85 ± 5 | 15 ± 8 | -5 ± 3 |
| Bevacizumab (100µg/mL) | VEGF-A Ligand | 75 ± 7 | 40 ± 10 | +20 ± 5* |
| Sunitinib | VEGFR/PDGFR | 90 ± 4 | 70 ± 6 | -30 ± 4 |
| Aflibercept | VEGF Trap (VEGF-A, PlGF) | 80 ± 6 | 55 ± 9 | +10 ± 4* |
*Positive value indicates increased core viability, suggesting worsened hypoxia.
Detailed Experimental Protocols
Protocol 1: Generating and Validating a Stable VEGF Gradient in a Microfluidic Device Objective: Create a linear, stable gradient to study endothelial cell migration. Materials: PDMS microfluidic chip (3-channel design), syringe pump, fluorescence-conjugated dextran (70kDa), time-lapse microscope. Steps:
- Chip Preparation: Sterilize chip with UV for 30 minutes. Coat central channel with 50µg/mL fibronectin for 1 hour.
- Gradient Generation: Load left reservoir with VEGF (100ng/mL) in serum-free media. Load right reservoir with serum-free media only. Set syringe pumps to a flow rate of 0.5µL/min for both input and output channels.
- Gradient Validation: Replace VEGF solution with FITC-dextran at the same concentration. After 1 hour, image the central channel using a 488nm laser. Plot fluorescence intensity across the channel width (ImageJ). A linear profile (R² > 0.95) indicates a stable gradient.
- Cell Experiment: Seed HUVECs (2x10⁴ cells) in the central channel. Allow adhesion for 4h. Initiate flow and gradient. Image migration every 15min for 12h.
Protocol 2: Isolating Stroma-Specific VEGF Signaling Feedback Objective: Decouple tumor-derived vs. stroma-derived VEGF signaling in vivo. Materials: Conditional Vegfa knockout mice (e.g., Vegfa^(fl/fl)), fibroblast-specific Cre mice (FSP1-Cre), tumor cell line (e.g., Lewis Lung Carcinoma), anti-CD31 antibodies. Steps:
- Mouse Model Generation: Cross Vegfa^(fl/fl) mice with FSP1-Cre mice to generate FSP1-Cre;Vegfa^(fl/fl) (stromal Vegfa KO) mice. Use Vegfa^(fl/fl) littermates as controls.
- Tumor Implantation: Inject 1x10⁶ LLC cells subcutaneously into both groups (n=8/group).
- Analysis:
- At 500mm³ volume, inject 60mg/kg pimonidazole i.p. 1h before sacrifice.
- Harvest tumors, section, and stain for: pimonidazole (hypoxia), CD31 (vasculature), α-SMA (CAFs), and Hoechst (nuclei).
- Quantify vessel density (CD31+ area/total area), perfusion (lectin injection), and hypoxic fraction (pimonidazole+ area/total area).
Diagrams
Diagram 1: Hypoxia-Driven VEGF Signaling Cascade in Tumors
Diagram 2: Experimental Workflow for Vasculature Heterogeneity Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Tumor Vasculature Heterogeneity Research
| Item | Function & Application | Example Product/Catalog # |
|---|---|---|
| Hypoxia Chamber / Workstation | Provides precise, controlled low-oxygen environment for cell culture. | Baker Ruskinn INVIVO2 400. |
| Pimonidazole HCl | Hypoxia probe. Forms adducts in cells at O₂ < 1.3%. Used for IHC/IF. | Hypoxyprobe-1 (HP1). |
| Recombinant Human VEGF-A165 | Gold-standard ligand for in vitro angiogenesis, migration, and gradient assays. | R&D Systems 293-VE. |
| VEGF ELISA Kit | Quantifies VEGF secretion from cells or tissue lysates. Critical for gradient validation. | Quantikine ELISA DVE00. |
| VEGFR-2 Tyrosine Kinase Inhibitor | Pharmacological blocker of primary VEGF signaling. Positive control for inhibition. | SU5416 (Semaxanib), Tocris 1476. |
| Fluorescent Lycopersicon Esculentum Lectin | In vivo perfusion marker. Binds selectively to perfused vasculature when injected intravenously. | Vector Laboratories DL-1178. |
| Anti-CD31 Antibody | Endothelial cell marker for visualizing and quantifying total tumor vasculature. | Abcam ab28364. |
| Microfluidic Chemotaxis Device | For establishing stable, quantifiable chemical gradients for migration studies. | Ibidi µ-Slide Chemotaxis 80326. |
| Matrigel (Growth Factor Reduced) | Basement membrane matrix for 3D culture and endothelial tubulogenesis assays. | Corning 356231. |
| HIF-1α Antibody | Western blot or IF detection of stabilized HIF-1α under hypoxia. | Novus Biologicals NB100-479. |
Technical Support Center
Troubleshooting Guide & FAQs
Q1: During intravital imaging of a subcutaneous tumor model, I observe significantly lower fluorescence signal from my vascular perfusion tracer (e.g., FITC-dextran) in the core region compared to the periphery. What could be the cause and how can I verify? A: This is a classic indicator of compromised and heterogeneous tumor vasculature. The core often has poorly functional, leaky, and immature vessels leading to reduced perfusion and increased interstitial fluid pressure.
- Troubleshooting Steps:
- Verify Tracer Properties: Ensure the dextran molecular weight (e.g., 70 kDa vs. 2 MDa) is appropriate. High MW tracers may extravasate less, highlighting permeability differences.
- Control for Injection: Standardize injection volume, rate, and site (e.g., tail vein). Inconsistent administration is a major source of variability.
- Co-stain for Vasculature: Perform immunofluorescence post-imaging on fixed tissue for CD31/PECAM-1. This will confirm if low signal is due to absence of vessels (necrosis) versus non-perfused vessels.
- Measure Hypoxia: Use a hypoxia probe (e.g., pimonidazole) injected prior to sacrifice. Hypoxic regions will spatially correlate with poorly perfused areas, confirming the biological consequence.
Q2: My flow cytometry data from dissociated tumors shows high variability in endothelial cell (CD31+) markers between samples. How can I improve the consistency of my stromal/vascular analysis? A: Variability often stems from inconsistent tissue sampling that fails to account for spatial zones.
- Troubleshooting Steps:
- Implement Spatial Sampling: Before dissociation, use a surgical blade or punch biopsy to physically separate the necrotic/core region (pale, soft) from the viable rim/periphery (pink, firm). Process and analyze these fractions separately.
- Use a Viability Dye: Include a robust live/dead dye (e.g., Zombie NIR) to exclude dead cells, which are abundant in the core and can non-specifically bind antibodies.
- Standardize Digestion: Use a multi-enzyme cocktail (see Protocol 1) and strictly control digestion time and temperature. Over-digestion degrades surface epitopes.
Q3: When establishing a "pre-metastatic niche" assay in the lung using conditioned media from tumor cells, my control mice also show mild inflammatory changes. How do I isolate the specific tumor-derived effect? A: This indicates potential non-specific effects from serum components or cellular stress products.
- Troubleshooting Steps:
- Employ Proper Controls: Include two critical controls alongside your tumor cell-conditioned media (TCM):
- Fresh Complete Media Control: Media incubated without cells for the same duration.
- Non-Neoplastic Cell Conditioned Media: e.g., from primary normal fibroblasts or untransformed epithelial cells.
- Fractionate TCM: Use centrifugal filters (e.g., 100 kDa, 10 kDa) to separate exosome/large vesicle fractions from soluble proteins. Test each fraction to identify the active component.
- Analyze Specific Markers: Move beyond general histology. Use flow cytometry or IHC for established pre-metastatic niche markers like CD11b+Ly6C+ myeloid-derived suppressor cells (MDSCs), LOX deposition, or S100A8/A9 upregulation to confirm tumor-specific education.
- Employ Proper Controls: Include two critical controls alongside your tumor cell-conditioned media (TCM):
Experimental Protocols
Protocol 1: Spatial Transcriptomic Profiling of Tumor Core vs. Periphery
- Objective: To compare gene expression profiles from the hypoxic, necrotic core and the normoxic, proliferative periphery of a solid tumor.
- Materials: Fresh tumor tissue (≥100 mg), OCT compound, cryostat, spatial transcriptomics slides (e.g., 10x Genomics Visium), dry ice, methanol.
- Method:
- Embed a freshly harvested, uncrosslinked tumor sample in OCT. Rapidly freeze on a dry ice/ethanol slurry or in liquid nitrogen-cooled isopentane. Store at -80°C.
- Section the tumor at 5-10 µm thickness using a cryostat. Collect sections directly onto the chilled areas of the spatial transcriptomics slide.
- Immediately fix slides in pre-chilled methanol at -20°C for 30 minutes. Air dry for 5 minutes.
- Stain with H&E according to the spatial platform's protocol and image the slide at high resolution.
- Perform tissue permeabilization, reverse transcription, and cDNA synthesis as per the manufacturer's instructions (e.g., 10x Visium User Guide).
- In the downstream computational analysis, manually annotate the core and periphery regions based on the H&E image (morphology, necrosis) and align these with the transcriptomic spots for comparative analysis.
Protocol 2: Isolation and Characterization of Endothelial Cells from Distinct Tumor Regions
- Objective: To obtain viable endothelial cells from spatially defined tumor regions for functional in vitro assays.
- Materials: Dissected tumor regions, digestion cocktail (Collagenase IV 1 mg/mL, Dispase II 1 U/mL, DNase I 20 µg/mL in PBS), FACS sorter, Endothelial Growth Medium (EGM-2).
- Method:
- Weigh the separately dissected core and peripheral tumor tissues.
- Mince each tissue separately with sterile scalpels into <1 mm³ pieces.
- Digest each sample in 5x volume of pre-warmed digestion cocktail for 30-45 minutes at 37°C with gentle agitation.
- Quench digestion with 10% FBS in PBS. Filter through a 70 µm cell strainer. Wash with PBS.
- Resuspend in FACS buffer (PBS + 2% FBS). Stain with anti-CD31-APC and anti-CD45-FITC antibodies for 30 min on ice. Include a viability dye (e.g., DAPI).
- Sort live (DAPI-), CD45-, CD31+ cells directly into EGM-2 medium using a FACS sorter.
- Culture sorted cells on fibronectin-coated plates in EGM-2. Assess functionality via tube formation assay on Matrigel, comparing core- vs. periphery-derived ECs.
Table 1: Comparative Metrics of Vasculature in Tumor Core vs. Periphery
| Metric | Tumor Core | Tumor Periphery | Measurement Technique | Reference Range (Typical Solid Tumor) |
|---|---|---|---|---|
| Vessel Density | Low | High | CD31 IHC, vWF staining | Core: 50-150 vessels/mm²; Periphery: 200-400 vessels/mm² |
| Perfusion Efficiency | Very Low (5-20%) | Moderate-High (40-70%) | FITC-dextran intravital imaging | Measured as % of CD31+ vessels containing tracer |
| Median pO₂ | Hypoxic (<5 mmHg) | Normoxic (~10-30 mmHg) | Hypoxyprobe, OxyLite probe | Core pO₂ often <1% of periphery |
| Vessel Maturity Index | Low (0.1-0.3) | Higher (0.4-0.7) | α-SMA+/CD31+ co-staining | Ratio of α-SMA+ mural cell-covered vessels to total vessels |
| Interstitial Fluid Pressure | High (15-40 mmHg) | Low-Moderate (5-10 mmHg) | Micropressure catheter | Can be 3-5x higher in core |
Table 2: Key Molecular Drivers in Metastatic Niche Formation
| Driver Molecule | Primary Source | Key Receptor/Target in Distant Organ | Functional Effect in Pre-Metastatic Niche | Common Assay for Detection |
|---|---|---|---|---|
| VEGF-A | Tumor cells, TAMs | VEGFR1/2 on endothelial & myeloid cells | Vascular permeability, immune cell recruitment | ELISA of plasma/serum; IHC |
| LOX / LOXL2 | Hypoxic tumor cells | Collagen IV, FN in ECM | ECM crosslinking, CD11b+ cell recruitment | Fluorescent LOX probe; IHC |
| S100A8/A9 | Myeloid cells, tumor cells | TLR4/RAGE on endothelial & resident cells | Pro-inflammatory signaling, cell adhesion | Flow cytometry (intracellular) |
| Exosomal miR-21 | Tumor-derived exosomes | TLR7/8 in resident macrophages | M2 macrophage polarization, immunosuppression | qPCR of exosomal RNA from plasma |
Diagrams
Title: Formation of the Pre-Metastatic Niche
Title: Spatial Heterogeneity Analysis Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Context | Example Product/Catalog # (for citation) |
|---|---|---|
| Hypoxyprobe (Pimonidazole HCl) | Binds covalently to proteins in hypoxic tissue (pO₂ < 10 mmHg). Essential for demarcating the necrotic/core region in IHC/IF. | Hypoxyprobe, Inc. (HP1-100Kit) |
| Lectin (e.g., Lycopersicon Esculentum) | Labels functional, perfused vasculature when injected intravenously prior to sacrifice. Contrasts with structural markers like CD31. | Vector Labs (DL-1174) |
| CD31/PECAM-1 Antibody | Gold-standard immunohistochemical marker for pan-endothelial cells to quantify vessel density and distribution. | BioLegend (102501); Abcam (ab28364) |
| α-SMA (Alpha-Smooth Muscle Actin) Antibody | Marks pericytes and vascular smooth muscle cells. Used with CD31 to calculate a vessel maturity index (core vs. periphery). | Sigma (A5228) |
| Collagenase IV / Dispase II Enzyme Cocktail | Optimized blend for gentle dissociation of tumor tissue to preserve endothelial cell surface markers for flow cytometry. | Worthington (LS004188 / LS02109) |
| Zombie NIR Fixable Viability Kit | Near-IR fluorescent dye for robust identification of dead cells in flow cytometry, critical for analyzing fragile cells from necrotic cores. | BioLegend (423105) |
| Matrigel Basement Membrane Matrix | Used for in vitro endothelial tube formation assays to compare the angiogenic potential of cells isolated from different regions. | Corning (356237) |
| Mouse Anti-S100A8/A9 Antibody | Detects key pro-inflammatory calprotectin heterodimer involved in pre-metastatic niche formation in lung/liver sections. | R&D Systems (MAB4576) |
Troubleshooting Guides & FAQs
Q1: In our liver metastasis model, we observe initial vessel co-option followed by rapid regression. Our anti-angiogenic therapy then fails. What is the likely mechanism and how can we adjust our protocol? A1: This pattern suggests a failed "Normalization" cycle. The regression phase is likely driven by intense anti-tumor immune response or excessive pruning by your therapeutic (e.g., VEGF inhibitor). The subsequent failure indicates a shift to a hypoxic, aggressive phenotype using alternative vascularization. Protocol Adjustment: Introduce a pulsed dosing schedule for the anti-angiogenic agent. Monitor with weekly CD31+ (pan-endothelial) and CD105+ (activated endothelial) dual immunohistochemistry to detect the narrow "normalization window" characterized by pericyte coverage and reduced vessel density. Administer your primary cytostatic drug during this window.
Q2: Our intravital microscopy data on vessel co-option dynamics are inconsistent. What are the critical controls for imaging live co-option in a cranial window? A2: Consistency requires strict control of physiological parameters. Essential Controls:
- Intracranial Pressure Control: Implement a saline reservoir to maintain constant pressure.
- Anesthesia Consistency: Use medically-grade isoflurane with a calibrated vaporizer; tail vein injections under ketamine/xylazine can alter hemodynamics.
- Temperature Homeostasis: Use a feedback-controlled heating pad set to 37°C for the mouse head stage.
- Vessel Labeling: Use a high-contrast, long-circulating agent like Dextran-FITC (2MDa). Always include a lectin (e.g., Lycopersicon Esculentum) injection post-sacrifice to label all perfused vasculature for final validation.
Q3: How do we quantitatively distinguish "vessel co-option" from "angiogenesis" in histology samples from heterogeneous tumors? A3: Use a multiplexed scoring approach on sequential sections or multiplex IF. Key differentiators are summarized in Table 1.
Table 1: Histological Discriminators of Co-option vs. Angiogenesis
| Feature | Vessel Co-option | Angiogenesis |
|---|---|---|
| Vessel Architecture | Normal, organ-typical pattern. | Dilated, tortuous, chaotic. |
| Vessel Maturity | High pericyte coverage (α-SMA+, NG2+). | Low or erratic pericyte coverage ("naked"). |
| Tumor-Vessel Interface | Tumor cells align along pre-existing basement membrane (Collagen IV+). | New basement membrane, often discontinuous. |
| Endothelial Proliferation | Low Ki67+ in endothelial cells. | High Ki67+ in endothelial cells. |
| Molecular Marker | Low VEGF-A expression; High Angiopoietin-1. | High VEGF-A, HIF-1α expression. |
Q4: Our "vessel normalization" therapy is causing excessive pruning and hypoxia in the tumor core. How do we titrate the dose? A4: You are likely beyond the therapeutic window. Implement a tiered dosing and monitoring protocol.
- Biomarker Monitoring: Measure circulating sVEGFR1 (sFlt1) and PlGF weekly. A rising PlGF/sFlt1 ratio indicates compensatory pathways.
- Imaging Gate: Use contrast-enhanced ultrasound (CEUS) to measure perfusion deficit. If >40% of tumor core becomes non-perfused, reduce dose by 50%.
- Histological Checkpoint: In a pilot cohort, sacrifice animals 72h after dose 3. Assess for hypoxia (pimonidazole adducts) and vessel density (CD31+). Aim for a 30-50% reduction in density without a significant increase in pimonidazole+ area.
Q5: What is the standard workflow to profile the complete "Co-option → Regression → Normalization" cycle in a single study? A5: Follow this integrated multi-modal workflow.
Diagram 1: Integrated workflow for profiling vascular cycles.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Vascular Dynamics Research
| Reagent / Material | Function & Application |
|---|---|
| Dextran, FITC, 2,000,000 MW | High molecular weight vascular label for intravital microscopy. Stays in circulation, defines perfused lumen. |
| DyLight Lycopersicon Esculentum Lectin | Binds to endothelial glycocalyx. Used as a definitive marker of all endothelial cells post-perfusion. |
| Hypoxyprobe (Pimonidazole HCl) | Forms protein adducts in hypoxic regions (<10 mmHg O2). Critical for quantifying therapy-induced hypoxia. |
| α-SMA (alpha-Smooth Muscle Actin) Antibody | Marker for pericytes and vascular smooth muscle. Key for assessing vessel maturity and normalization. |
| CD31/PECAM-1 Antibody (Clone SZ31) | Pan-endothelial cell marker for immunohistochemistry and flow cytometry. Best for vessel density quantification. |
| CD105/Endoglin Antibody | Marks activated/proliferating endothelial cells. Differentiates angiogenic sprouts from co-opted vessels. |
| Matrigel GFR, Phenol Red-Free | For in vitro endothelial tube formation assays to test tumor-secreted factor activity. Use low-growth factor for purity. |
| sVEGFR1 (sFlt-1) ELISA Kit | Measures circulating biomarker of anti-angiogenic response and vascular stress. |
Technical Support Center
Welcome to the Technical Support Center for research on tumor vasculature heterogeneity. This guide provides troubleshooting and FAQs for experimental challenges related to poor perfusion, increased interstitial fluid pressure (IFP), and drug resistance.
Troubleshooting Guide & FAQs
Q1: Our in vivo imaging shows heterogeneous and poor perfusion of the fluorescently labeled therapeutic antibody. How can we verify this quantitatively and identify hypoxic regions?
- A: Heterogeneous perfusion is a hallmark of abnormal tumor vasculature. To quantify:
- Dynamic Contrast-Enhanced MRI (DCE-MRI): Use a gadolinium-based contrast agent. The key quantitative parameter is Ktrans (volume transfer constant), which reflects perfusion and vascular permeability. Low Ktrans indicates poor perfusion.
- Hypoxia Staining: Administer pimonidazole (60 mg/kg, i.p.) 60-90 minutes before tumor excision. Fix tissue, section, and immunostain with an anti-pimonidazole antibody (e.g., Hypoxyprobe). Co-stain with CD31 to correlate hypoxia with vasculature.
Q2: We are measuring Interstitial Fluid Pressure (IFP) in murine xenografts, but our readings are inconsistent. What is the best practice?
- A: High IFP is a major barrier to drug delivery. Use the "gold-standard" wick-in-needle or micropressure catheter system (e.g., Millar).
- Protocol: Anesthetize the mouse and stabilize the tumor. Carefully insert the calibrated needle/catheter into the tumor core. Avoid necrotic areas (soft regions) and major vessels. Record the pressure once a stable plateau is reached (≈2-3 min). Take multiple measurements (n≥5) across different tumor regions. Always include a normal tissue control (e.g., muscle).
- Troubleshooting: Inconsistent readings often stem from needle clogging or placement in necrotic zones. Verify system calibration with a water column before each session.
Q3: Our drug is effective in 2D culture but fails in 3D spheroids and in vivo models. Could high IFP and poor penetration be the cause?
- A: Very likely. This is a classic sign of physical drug resistance. Implement a spheroid penetration assay.
- Protocol:
- Generate uniform spheroids (∼500µm diameter) using U-bottom plates or hanging drop method.
- Treat spheroids with a fluorescently tagged version of your drug (or a surrogate dye of similar size/charge).
- At set timepoints (e.g., 6h, 24h), image spheroids using confocal microscopy with z-stacking.
- Quantify fluorescence intensity from the rim to the core. Poor penetration will show a steep gradient.
- Protocol:
Q4: Which signaling pathways should we target to normalize tumor vasculature and potentially improve perfusion and reduce IFP?
- A: The VEGF pathway is primary, but others are crucial.
Key Pathways in Vascular Abnormalities & Normalization
Q5: What are the key quantitative metrics to document when studying these barriers?
- A: Use the table below to standardize your reporting.
| Metric | Technique | Typical Value (Tumor vs. Normal) | Indicates |
|---|---|---|---|
| Perfusion (Ktrans) | DCE-MRI | Tumor: 0.05-0.15 min⁻¹, Normal: >0.5 min⁻¹ | Low = Poor, heterogeneous delivery |
| Interstitial Fluid Pressure (IFP) | Micropressure Catheter | Tumor: 10-40 mmHg, Normal: 0-3 mmHg | High = Reduced convection, barrier |
| Hypoxic Fraction | Pimonidazole IHC | Tumor: 10-50%, Normal: ~0% | Regions of therapeutic resistance |
| Drug Penetration Depth | Spheroid Imaging | Gradient over 50-200 µm | Steep gradient = Poor penetration |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Experiment |
|---|---|
| Pimonidazole HCl (Hypoxyprobe) | Forms protein adducts in hypoxic tissues (<10 mmHg O₂); detected via IHC to map tumor hypoxia. |
| Fluorescent Dextrans (e.g., 70 kDa FITC-dextran) | Vascular permeability and perfusion tracer; used to quantify extravasation and blood flow in vivo. |
| Anti-CD31/PECAM-1 Antibody | Endothelial cell marker for immunohistochemistry to visualize and quantify tumor blood vessel density. |
| Recombinant VEGF / Anti-VEGF Antibody | To stimulate (VEGF) or inhibit (antibody) vascular abnormalities in perturbation studies. |
| Gadoteridol / Gadobutrol | MRI contrast agents for non-invasive, quantitative DCE-MRI to calculate Ktrans. |
| Pressure Catheter (Millar SPR-1000) | Direct, precise measurement of interstitial fluid pressure in solid tumors. |
Experimental Protocol: Integrated Assessment of Vascular Barriers
Workflow for Multi-Parameter Vascular Analysis
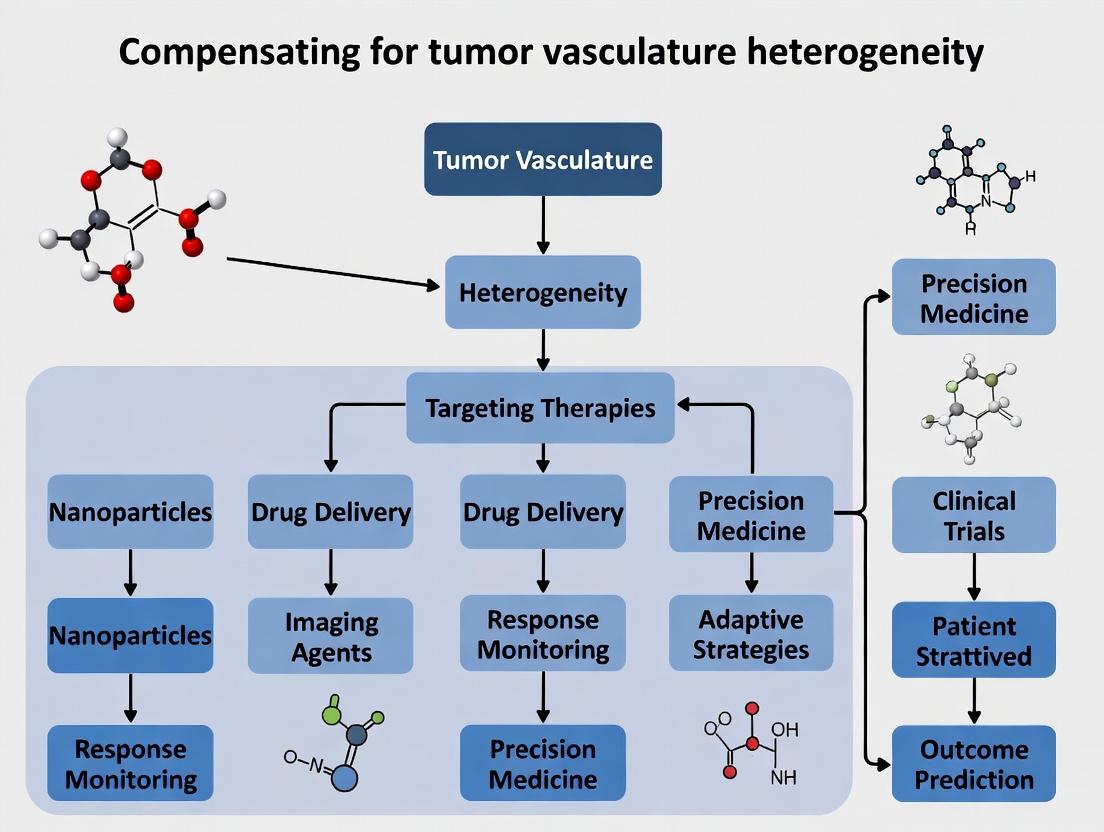
Bridging the Gaps: Methodologies to Homogenize Delivery and Targeting
Troubleshooting Guides and FAQs
Q1: Our anti-angiogenic treatment is failing to improve drug delivery in our xenograft model, and sometimes even reduces perfusion. What are the primary causes? A: This is a classic sign of over-pruning of the vasculature, pushing it from a "normalized" state to an overly regressed one. Key factors include:
- Excessive Dosing: The anti-angiogenic dose is too high. The goal is moderation, not maximal vessel destruction.
- Incorrect Timing: The therapeutic window of vascular normalization is transient. Administering your chemotherapy or immunotherapy outside this window (too early or too late) misses the opportunity.
- Lack of Biomarker Monitoring: Proceeding without verifying normalization via biomarkers means you are operating blind. Solution: Implement the Experimental Protocol 1: Dynamic Biomarker Assessment below to guide dosing schedules.
Q2: Which biomarkers are most reliable for identifying the vascular normalization window in real-time? A: No single biomarker is perfect. A combination is required for robust assessment. Quantitative data from recent studies is summarized in Table 1.
Table 1: Key Biomarkers for Vascular Normalization
| Biomarker Category | Specific Marker | Normalization Trend | Measurement Technique | Key Insight |
|---|---|---|---|---|
| Structural | Pericyte Coverage (α-SMA) | Increases | IHC, IF | Aim for ~70-80% coverage; low coverage indicates immaturity, very high may indicate over-stabilization. |
| Functional | Tumor Hypoxia (pimonidazole) | Decreases transiently | IHC | A initial decrease indicates improved perfusion; a subsequent rise signals over-pruning and renewed hypoxia. |
| Molecular | Plasma VEGF-A | Decreases | ELISA | Steady decline often correlates with response. A sudden spike may indicate compensatory resistance. |
| Molecular | SDF1α / Ang2 Ratio | Increases | Multiplex ELISA | A higher ratio is associated with a pro-normalization microenvironment. |
| Imaging | Ktrans (DCE-MRI) | Increases then plateaus | DCE-MRI | An initial increase in perfusion/permeability indicates normalization. A drop below baseline signals over-treatment. |
Q3: How do we determine the optimal biological dose (OBD) for normalization, as opposed to the maximum tolerated dose (MTD)? A: The OBD is defined by the peak of the normalization window, not toxicity. Follow Experimental Protocol 2: OBD Determination below. It requires a multi-parameter approach where improved perfusion (e.g., Ktrans), reduced hypoxia, and increased pericyte coverage are plotted against dose. The OBD is where these parameters are optimally improved before worsening.
Q4: We see high inter-tumor and intra-tumor heterogeneity in biomarker response. How should we adapt our protocol? A: This is a core challenge in compensating for vasculature heterogeneity.
- Sampling: Take multiple core biopsies from different tumor regions (edge vs. core) for IHC analysis.
- Imaging: Use whole-tumor imaging modalities (DCE-MRI, PAT) to capture spatial heterogeneity.
- Adaptive Dosing: Consider biomarker-driven adaptive trials where the dose or schedule is modified based on early biomarker signals from the individual tumor.
Experimental Protocols
Experimental Protocol 1: Dynamic Biomarker Assessment for Scheduling
Objective: To empirically determine the normalization window for scheduling combination therapies.
- Model Establishment: Implant tumors (e.g., murine MC38 or 4T1).
- Treatment Initiation: Administer a candidate anti-angiogenic agent (e.g., anti-VEGFR2 antibody DC101 at 10-20 mg/kg).
- Longitudinal Monitoring:
- Day 3, 5, 7, 10, 14: Image a cohort of mice via DCE-MRI to calculate Ktrans.
- Day 5 & 9: Inject pimonidazole (60 mg/kg, i.p.) 90 min before sacrifice. Harvest tumors.
- Analysis: Process tumors for IHC: CD31 (endothelium), α-SMA (pericytes), pimonidazole (hypoxia).
- Determine Window: The normalization window is typically the period where Ktrans is elevated, hypoxia is minimized, and pericyte coverage is increased. Schedule your combination therapy (chemotherapy or anti-PD-1) within this period.
Experimental Protocol 2: Determining the Optimal Biological Dose (OBD)
Objective: To find the dose that maximizes vascular normalization.
- Dose Cohorts: Establish groups receiving a range of doses (e.g., 5, 10, 20, 40 mg/kg of a tyrosine kinase inhibitor like sunitinib).
- Endpoint Analysis: Treat for one week. On Day 7, perform DCE-MRI, inject pimonidazole, and harvest tumors.
- Multi-Parametric Scoring: Quantify for each dose: (a) Mean Ktrans, (b) Hypoxic fraction (%), (c) Pericyte coverage index (α-SMA+ area / CD31+ area).
- Plot & Identify: Plot each parameter (normalized to control) against the dose. The OBD is the dose where the composite benefit peaks before declining.
Pathway and Workflow Diagrams
Title: Anti-VEGF Therapy Dose Response Pathways
Title: Biomarker-Guided Therapy Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Vascular Normalization Studies
| Item | Function & Rationale |
|---|---|
| Anti-VEGFR2 Antibody (e.g., DC101) | The gold-standard research tool for selectively blocking mouse VEGFR2 to induce vascular pruning/normalization in syngeneic models. |
| Pimonidazole HCl | Hypoxia probe. Forms protein adducts in cells with pO₂ < 10 mm Hg, detectable by IHC/IF, allowing quantification of tumor hypoxia. |
| Fluorescent/Dextran Conjugates (e.g., FITC-Dextran) | Used for vessel perfusion studies. Injected intravenously; extravasation and distribution visualize functional vasculature and permeability. |
| Phospho-Specific Antibodies (p-VEGFR2, p-Akt) | For assessing pathway inhibition/activation in endothelial cells within the tumor stroma via IHC. |
| Multiplex ELISA Panel (Mouse) | For simultaneous measurement of key circulating cytokines (VEGF, PlGF, SDF1α, Ang2) from small-volume plasma samples to monitor systemic response. |
| α-SMA & NG2 Antibodies | For identifying pericytes and quantifying pericyte coverage on tumor vessels (CD31+ structures). |
| Tyrosine Kinase Inhibitors (e.g., Sunitinib, Pazopanib) | Small molecule multi-target inhibitors used to study the effects of broader pathway inhibition compared to specific antibody blockade. |
Technical Support Center
Frequently Asked Questions (FAQs)
Q1: Our in vivo tumor permeability assay shows high variability after STING agonist administration. What are the critical control points? A1: Key control points include:
- Agent Preparation: Ensure STING agonist (e.g., DMXAA, cGAMP) is reconstituted in the correct vehicle (e.g., sterile PBS, 5% DMSO/saline) and administered at a consistent time in the tumor growth cycle.
- Timing of Measurement: Permeability peaks 4-24 hours post-administration depending on the agonist and tumor model. Conduct a pilot time-course experiment.
- Vascular Labeling: Use a consistent, high-purity fluorescent dextran (e.g., 70 kDa FITC-dextran) and a standardized circulation time (typically 3-10 minutes) before perfusion and fixation.
Q2: When combining radiotherapy with permeability-enhancing agents, how do we sequence the treatments? A2: Sequence is critical. For a synergistic effect aimed at enhancing drug delivery:
- Administer the permeability-enhancing agent (e.g., a STING agonist).
- Wait for the predicted peak in vascular normalization/permeability (e.g., 24 hours post-STING agonist).
- Apply radiotherapy (e.g., a single 8 Gy fraction).
- Administer the primary therapeutic agent (e.g., chemotherapy, antibody-drug conjugate) within the subsequent 4-24 hour window. Always include control groups for each treatment alone and the reverse sequence.
Q3: Our immunofluorescence staining for endothelial markers (CD31) is weak or inconsistent after radiotherapy. How can we improve this? A3: This is common due to radiation-induced endothelial damage.
- Antigen Retrieval: Use a high-temperature, high-pressure citrate-based antigen retrieval step for 15-20 minutes.
- Antibody Validation: Confirm your anti-CD31 antibody clone is validated for your specific tumor model (mouse/human) and for use on irradiated tissue.
- Alternative Markers: Consider co-staining with a pan-endothelial marker like ERG or von Willebrand Factor (vWF) for confirmation.
Q4: What is the best method to quantitatively assess permeability changes in heterogenous tumors? A4: Use a multi-modal approach:
- Dynamic Contrast-Enhanced MRI (DCE-MRI): Provides in vivo, spatially resolved quantitative data (Ktrans) across the entire tumor. Correlate with ex vivo analysis of specific regions.
- Ex vivo Fluorescence Microscopy: After injecting a fluorescent tracer in vivo, excise the tumor, section it, and quantify extravasated tracer intensity relative to vascular area (using CD31 staining) in multiple, defined regions (core, periphery).
- Evans Blue Dye Assay: A simple, quantitative bulk measurement of total tumor permeability for validation.
Troubleshooting Guides
Issue: Lack of Expected Permeability Increase with STING Agonist.
| Symptom | Possible Cause | Solution |
|---|---|---|
| No change in dextran extravasation. | Inactive agonist batch; incorrect dosage; non-responsive tumor model. | Validate agonist activity in a reporter cell assay (e.g., THP1-Dual cells). Titrate dose. Check literature for model responsiveness (e.g., B16 vs. 4T1). |
| Increased permeability but excessive necrosis. | Dosage too high, causing severe vascular damage. | Reduce dose by 50-75% and monitor for a "normalization window." |
| High animal-to-animal variability. | Subcutaneous tumor size/volume disparities. | Standardize tumor volume at treatment initiation (e.g., 100-150 mm³). |
Issue: Inconsistent Radiotherapy Effects on Tumor Vasculature.
| Symptom | Possible Cause | Solution |
|---|---|---|
| No vascular changes post-radiation. | Incorrect dose calculation or field placement. | Calibrate irradiator source. Use CT-guided or precision conformal radiotherapy to ensure entire tumor is targeted. |
| Excessive vascular collapse, hindering drug delivery. | Single dose too high. | Consider fractionated dosing (e.g., 3 x 3 Gy) to promote a more sustained normalization phenotype. |
| Cannot correlate permeability with immune cell influx. | Lack of spatial registration in analysis. | Use serial tissue sections for CD31 (vessels), dextran (permeability), and CD8 (T-cell) staining. Employ image analysis software to co-localize signals. |
Table 1: Comparative Effects of Permeability-Enhancing Strategies
| Strategy | Typical Dose/Regimen | Peak Effect Onset | Key Metric Change (vs. Control) | Common Tumor Models |
|---|---|---|---|---|
| STING Agonist (cGAMP) | 50 µg intratumoral | 12-24 hours | Ktrans (MRI): +150-200% | 4T1, MC38, B16F10 |
| STING Agonist (DMXAA) | 25 mg/kg i.p. | 4-8 hours | Dextran (70 kDa) Extravasation: +300% | CT26, LLC |
| Radiotherapy (Single Dose) | 8 Gy, focal | 1-3 days | Ktrans: Initial ↓ (0-1d), then ↑ (1-3d) | GL261, U87, HNSCC PDX |
| Fractionated Radiotherapy | 3 x 3 Gy, daily | Sustained over course | Vascular Density (CD31+): Normalized (+10-20%) | Various PDX models |
| Combinatorial (STING + RT) | cGAMP (50 µg) + 8 Gy (24h later) | 48h post-STING | Drug Delivery (Doxorubicin): +400% | 4T1, EMT6 |
Table 2: Key Reagents for Assessing Vascular Permeability
| Reagent | Target/Function | Example Product Code | Critical Application Note |
|---|---|---|---|
| FITC- or TRITC-Dextran | Vascular tracer for permeability. | D1822, D1818 (Thermo Fisher) | Use 70 kDa for physiologic permeability; 2000 kDa for gross leakage. |
| Anti-CD31 Antibody | Platelet endothelial cell adhesion molecule (PECAM-1) for vessel staining. | 550274 (BD Biosciences) | Optimal for mouse tissue. Validate clone for irradiated samples. |
| Anti-Collagen IV Antibody | Basement membrane marker for vascular integrity. | AB769 (Millipore) | Co-stain with CD31 to assess vessel maturity/normalization. |
| Recombinant STING Agonist | Activates the STING pathway in immune/endothelial cells. | tlrl-cga (InvivoGen) | For mouse cells. Aliquot to avoid freeze-thaw cycles. |
| Evans Blue Dye | Albumin-binding dye for gross permeability quantification. | E2129 (Sigma-Aldrich) | Circulate for 30 min. Extract dye with formamide at 55°C for 24h. |
Experimental Protocols
Protocol 1: Ex Vivo Quantitative Tumor Vascular Permeability Assay Objective: To measure the extravasation of a fluorescent tracer from tumor blood vessels.
- Tracer Injection: Anesthetize tumor-bearing mouse. Inject 100 µL of 10 mg/mL FITC-labeled 70 kDa dextran (in PBS) via the tail vein.
- Circulation: Allow the tracer to circulate for exactly 5 minutes.
- Vascular Perfusion: Without delay, perfuse the mouse transcardially with 20 mL of PBS followed by 20 mL of 4% paraformaldehyde (PFA) at a constant pressure (120 mmHg) to clear intravascular dextran.
- Tumor Harvest: Excise the tumor and post-fix in 4% PFA for 4 hours at 4°C, then transfer to 30% sucrose for 48 hours for cryoprotection.
- Imaging: Embed tumor in OCT, section at 50 µm. Image entire sections using a fluorescence microscope with consistent settings.
- Analysis: Threshold images for FITC signal. Co-stain with anti-CD31 antibody on a serial section. Calculate the ratio of extravascular (CD31-negative) FITC area to total CD31-positive area per field across multiple tumor regions.
Protocol 2: Combining STING Agonist with Radiotherapy for Enhanced Permeability Objective: To schedule treatments to maximize therapeutic delivery window.
- Day 0: Implant tumor cells subcutaneously.
- Day 10-12: When tumors reach ~100 mm³, randomize mice into groups (Control, STING only, RT only, STING+RT).
- Day 1 (Treatment): STING+RT group receives intratumoral injection of cGAMP (e.g., 50 µg in 30 µL PBS).
- Day 2: 24 hours post-STING injection, anesthetize mice in the RT and STING+RT groups. Shield body and deliver focal radiotherapy (e.g., 8 Gy) to the tumor using a small animal irradiator.
- Day 2.5: 4-6 hours post-RT, administer your primary therapeutic agent (e.g., chemotherapy, nanoparticle).
- Day 3: 24 hours post-therapeutic agent, assess permeability using Protocol 1 or harvest tumors for analysis of drug penetration.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Research |
|---|---|
| Fluorescent Dextrans (Various Sizes) | Macromolecular tracers to simulate drug/antibody leakage from vasculature. |
| Anti-CD31/PECAM-1 Microbeads | For isolating primary tumor endothelial cells for ex vivo analysis of pathway activation. |
| STING Reporter Cell Line (e.g., THP1-Dual) | To quantitatively validate the activity of STING agonist batches via secreted luciferase. |
| Hypoxyprobe (Pimonidazole HCl) | To identify hypoxic regions within the tumor, which correlate with poor perfusion and drug delivery. |
| Matrigel Plug Assay Kit | In vivo assay to study angiogenesis and compound effects on vessel formation and permeability. |
| Mouse Anti-Collagen IV, Laminin Antibodies | To assess vascular basement membrane thickness and integrity, indicators of normalization. |
Visualizations
STING Pathway Leads to Enhanced Permeability
STING & Radiotherapy Combination Workflow
Technical Support Center: Troubleshooting & FAQs
This support center is framed within the thesis research context of "Compensating for Tumor Vasculature Heterogeneity to Improve Nanocarrier Delivery via the Enhanced Permeation and Retention (EPR) Effect." It addresses common experimental challenges in designing nanocarriers to overcome variable and inefficient tumor vascularization.
Frequently Asked Questions (FAQs)
Q1: Our polymeric nanoparticles (100 nm spherical) show high accumulation in a subcutaneous xenograft model but poor performance in an orthotopic or metastatic model. Is this a size issue? A: Likely not solely a size issue. Heterogeneous vasculature between tumor models is a key factor. Subcutaneous tumors often have more uniform, well-developed vasculature compared to orthotopic/metastatic sites, which better mimic human tumor heterogeneity. The "average" 100 nm size may be optimal for homogeneous vasculature but fail in heterogeneous pores. Consider a polydisperse system or co-administering a vessel-normalizing agent (e.g., anti-VEGF) to "standardize" the vascular pore size.
Q2: We engineered rod-shaped particles for improved margination and penetration, but in vivo tracking shows they accumulate primarily in the liver and spleen, not the tumor. What went wrong? A: This is a classic shape-mediated clearance issue. While rods may exhibit favorable hemodynamics, aspect ratios >3-5 are often efficiently phagocytosed by macrophages in the reticuloendothelial system (RES). Your surface engineering likely does not compensate for the shape-dependent clearance. Implement a denser PEGylation regimen or use a macrophage "don't eat me" signal (e.g., CD47 mimetic peptides) on the surface.
Q3: We see batch-to-batch variation in tumor accumulation even with identical nanoparticle synthesis protocols. Could surface charge be the variable? A: Yes. Minute changes in surface charge (zeta potential) significantly impact protein corona formation, which dictates biological identity. A shift from -15 mV to -5 mV can lead to rapid opsonization and clearance. Rigorously monitor and control zeta potential. Consider formulating at a slightly negative charge (-10 to -20 mV) for reduced non-specific interaction, but validate for your specific polymer/lipid system.
Q4: Does actively targeting nanoparticles (e.g., with folate or RGD peptides) truly improve extravasation in heterogeneous EPR, or just internalization? A: Primarily internalization. Active targeting ligands enhance receptor-mediated cellular uptake after the particle has extravasated through vascular pores. They do not significantly aid the initial passive extravasation step through heterogeneous pores. In fact, excessive targeting can increase clearance. Use moderate ligand density (1-5% molar ratio) to avoid masking the stealth coating while retaining binding capability post-extravasation.
Q5: Our nanoparticles work in murine models but fail in preliminary primate studies. Is the EPR effect not translational? A: The EPR effect is real in humans but is markedly more heterogeneous than in commonly used murine models. Murine tumors are often fast-growing with uniform, leaky vasculature. Human tumors are more complex, with denser stroma and variable perfusion. Your nanocarrier design must account for this by incorporating strategies like:
- Size-tuning: Develop a library of particles (70-150 nm) to target a range of pore sizes.
- Physicochemical responsiveness: Use pH- or enzyme-sensitive shields that only "activate" in the tumor.
- Adjuvant therapies: Combine with radiotherapy or photodynamic therapy to enhance local vascular permeability.
Troubleshooting Guides
Issue: Low Tumor Accumulation Despite Optimal In Vitro Characterization
- Check 1: Validate the EPR Effect in Your Model. Measure tumor vascular permeability using fluorescent dextrans of varying sizes (e.g., 70 kDa FITC-dextran ~12 nm). If dextrans don't accumulate, EPR is weak.
- Check 2: Protein Corona Analysis. Isolate nanoparticles from blood plasma ex vivo and analyze adsorbed proteins via SDS-PAGE or LC-MS. A corona rich in opsonins (e.g., immunoglobulins, complement) explains rapid clearance.
- Check 3: Pharmacokinetics/Biodistribution. Ensure you are taking early time points (1-4 hrs). Some nanoparticles have a rapid distribution phase and may localize to tumors earlier than the standard 24-hr endpoint.
Issue: High Tumor Accumulation but Low Therapeutic Efficacy
- Check 1: Intratumoral Distribution. Use fluorescence microscopy/IHC. Your nanoparticles may be trapped perivascularly and not penetrate the hypoxic, high-pressure tumor core. Solution: Reduce size (<50 nm) or incorporate enzymatic degradation motifs.
- Check 2: Drug Release Kinetics. The tumor microenvironment may not trigger release effectively. Compare release profiles in in vitro vs. simulated tumor conditions (mild acidity, specific enzymes). Implement dual-sensitive triggers.
- Check 3: Off-target Effects. Check for premature drug leakage in circulation, which causes toxicity and reduces payload delivery.
Experimental Protocols for Key Cited Experiments
Protocol 1: Assessing Tumor Vasculature Heterogeneity via Multisize Dextran Profiling Objective: To characterize the functional pore size distribution in a specific tumor model. Materials: See "Research Reagent Solutions" table. Method:
- Prepare solutions of fluorescent dextrans (e.g., 10 kDa, 40 kDa, 70 kDa, 150 kDa) in PBS.
- Inject each dextran separately (or as a cocktail if using distinct fluorophores) intravenously into tumor-bearing mice (n=5 per group).
- At a fixed time point (e.g., 30 minutes post-injection), euthanize and perfuse with saline to clear intravascular dye.
- Excise tumors and homogenize. Quantify fluorescence in homogenate supernatant.
- Image tumor sections via confocal microscopy to visualize spatial distribution.
- Data Analysis: Calculate % injected dose per gram (%ID/g) for each dextran. The ratio of accumulation of large vs. small dextrans indicates pore size heterogeneity.
Protocol 2: Evaluating the Impact of Shape on Circulation and Tumor Targeting Objective: To compare the pharmacokinetics of spherical vs. rod-shaped nanocarriers. Method:
- Synthesize spherical and rod-shaped nanoparticles (e.g., from PLGA or mesoporous silica) with identical surface chemistry (PEG, charge).
- Label both formulations with a near-infrared dye (e.g., DiR).
- Inject formulations into separate groups of mice (n=6-8) via tail vein.
- Collect blood samples at serial time points (5 min, 30 min, 2 hr, 8 hr, 24 hr).
- Euthanize at terminal time point (24 hr), harvest major organs and tumors.
- Measure fluorescence in blood and tissue lysates to determine blood half-life and biodistribution.
- Key Metric: Compare the area under the curve (AUC) for blood concentration and the tumor-to-liver ratio.
Protocol 3: Testing a Heterogeneity-Compensating "Mixed-Population" Strategy Objective: To determine if a cocktail of different-sized nanoparticles improves overall delivery. Method:
- Prepare three distinct, color-coded nanoparticle batches: Small (S, 30 nm), Medium (M, 100 nm), Large (L, 150 nm). Use different fluorophores (e.g., Cy5, Cy7, IR800).
- Inject groups of mice with: a) S only, b) M only, c) L only, d) a cocktail of S+M+L.
- Perform longitudinal in vivo imaging at 1, 4, 8, 12, 24 hours.
- At 24 hours, quantify signal in tumors and RES organs ex vivo.
- Analysis: The cocktail group should show more consistent and higher total tumor fluorescence across individual mice, indicating compensation for varying pore sizes.
Data Presentation
Table 1: Impact of Nanocarrier Size on Pharmacokinetic and Tumor Accumulation Parameters in Heterogeneous Tumor Models
| Parameter | Small (30 nm) | Medium (100 nm) | Large (150 nm) | Optimal for Heterogeneous EPR |
|---|---|---|---|---|
| Blood Half-life (hr) | 8.2 ± 1.5 | 12.5 ± 2.1 | 4.3 ± 0.9 | Medium |
| Tumor %ID/g (24 hr) | 3.5 ± 1.2 | 6.8 ± 3.5 | 2.1 ± 0.8 | Medium (but high variance) |
| Liver %ID/g (24 hr) | 18.5 ± 3.1 | 25.2 ± 4.7 | 35.8 ± 6.2 | Small |
| Penetration Depth (μm from vessel) | 80 ± 25 | 40 ± 15 | 15 ± 10 | Small |
| Interpretation | Good penetration, low RES uptake but moderate tumor accumulation. | Best tumor accumulation on average, but poor penetration and high liver uptake. | Rapid clearance, poor tumor access. | A cocktail of Small + Medium may yield optimal coverage. |
Table 2: Surface Engineering Strategies to Mitigate Heterogeneity Challenges
| Strategy | Typical Implementation | Effect on Circulation | Effect on Tumor Targeting | Risk in Heterogeneous Vasculature |
|---|---|---|---|---|
| PEGylation (Stealth) | 5 kDa PEG, 5-10% molar density | ++ (Greatly Increased) | + (Passive, via EPR) | Low. The gold standard for reducing clearance. |
| Charge Shielding | Zwitterionic polymers (e.g., PCB) | +++ (Very High) | + (Passive, via EPR) | Low. Excellent for reducing protein adsorption. |
| Active Targeting | Folate, RGD peptides (1-2% density) | - (Can decrease) | ++ (Increased cellular uptake post-extravasation) | High if density is too high, leads to RES recognition. |
| Stimuli-Responsive Shedding | PEG linked via MMP-9 cleavable peptide | ++ (Long, then shed) | +++ (Exposes binding motifs in tumor) | Medium. Depends on reliable enzyme expression in target area. |
| Biomimetic Coating | Leukocyte or RBC membrane coating | +++ (Very High) | + to ++ (Can have active targeting) | Low to Medium. Highly complex but promising. |
The Scientist's Toolkit: Research Reagent Solutions
| Item (Supplier Example) | Function in Heterogeneous EPR Research |
|---|---|
| Fluorescent Dextrans (Sigma-Aldrich) | Polysaccharide probes of defined size (e.g., 70 kDa = ~12 nm) to map functional vascular pore sizes. |
| DSPE-PEG(2000) (Avanti Polar Lipids) | Gold-standard PEG lipid for creating stealth coatings on liposomes and polymeric nanoparticles. |
| PLGA (Evonik) | Biodegradable, FDA-approved copolymer for forming size-controlled nanoparticles via nanoprecipitation. |
| MMP-9 Substrate IV (Calbiochem) | Peptide sequence (GPLGIAGQ) used to create enzyme-sensitive linkers for tumor-specific deshielding. |
| Anti-CD31 Antibody (BioLegend) | For immunohistochemical staining of tumor blood vessels to assess vascular density and morphology. |
| Near-IR Dyes (e.g., DiR, Li-Cor) | For in vivo and ex vivo optical imaging of nanoparticle biodistribution and pharmacokinetics. |
| Dynamic Light Scattering (DLS) Instrument | For critical measurement of nanoparticle hydrodynamic diameter, PDI, and zeta potential. |
| Orthotopic Tumor Model Cell Lines | Tumor cells engineered for luciferase expression to enable implantation and growth in native organ sites, providing more heterogeneous vasculature models. |
Mandatory Visualizations
Title: Nanocarrier Design Strategies to Overcome Vascular Heterogeneity
Title: Experimental Workflow for Heterogeneity-Targeted Nanocarrier Evaluation
Title: Protein Corona Formation Dictates Nanocarrier Fate In Vivo
Vascular Disruption Agents (VDAs) and their Controlled Application
Technical Support Center: Troubleshooting and FAQs
This support center is designed to assist researchers working within the thesis framework of Compensating for Tumor Vasculature Heterogeneity. It addresses common experimental challenges with VDA application.
Frequently Asked Questions (FAQs)
Q1: Our in vivo model shows extreme variability in VDA response between tumors, even from the same cell line. How can we account for this in our experimental design? A: This variability is a direct manifestation of tumor vasculature heterogeneity. To compensate:
- Pre-stratify animals using dynamic contrast-enhanced MRI (DCE-MRI) or contrast-enhanced ultrasound (CEUS) to quantify baseline perfusion parameters (e.g., Ktrans, blood volume) before VDA administration.
- Group animals by perfusion phenotype (e.g., high vs. low), not just tumor volume.
- Increase cohort size to account for intrinsic vascular heterogeneity. A power analysis based on your preliminary perfusion data is essential.
Q2: Following CA4P (Fosbretabulin) administration, we observe a robust central necrotic response but subsequent rapid peripheral regrowth. What strategies can mitigate this? A: This is a classic limitation due to the surviving viable rim, fed by heterogeneous, often normalized, vasculature. Consider these combination strategies:
- Sequence with anti-angiogenics (e.g., Bevacizumab): Administer after VDA to prune the reactive vessels in the rim, potentially prolonging VDA effect.
- Combine with cytotoxic chemotherapy: Schedule chemotherapy (e.g., Doxorubicin) 18-24 hours post-VDA, during the window of enhanced drug delivery to the rim.
- Pair with radiation therapy: Target the oxygenated, metabolically active rim shortly after VDA treatment.
Q3: What is the optimal time window for administering a secondary therapy (e.g., chemotherapy) after a VDA dose? A: The optimal window is typically narrow and agent-dependent. For tubulin-binding VDAs like CA4P:
- Peak vascular shutdown occurs at ~1-6 hours post-administration.
- The "therapeutic window" for enhanced delivery to the viable rim is generally between 18 to 72 hours, as vasoconstriction subsides and blood flow is partially re-established in remaining vessels. Empirical validation in your specific model is critical.
Q4: How do we differentiate between true vascular shutdown and transient vascular stasis in our imaging assays? A: Utilize multi-parametric imaging:
- Perfusion Imaging (DCE-MRI/CEUS): Measures blood flow and volume. A persistent drop (>24h) indicates shutdown.
- Diffusion-Weighted MRI (DW-MRI): Monitors cellular density. Increased apparent diffusion coefficient (ADC) values follow necrosis induced by successful shutdown.
- Histology Correlation: Use dual-endothelial cell (CD31) and hypoxia (pimonidazole) staining on terminal tissues. True shutdown leads to widespread endothelial cell disruption and hypoxia.
Troubleshooting Guides
Issue: Lack of Expected Antitumor Efficacy with a Promising VDA Candidate
| Possible Cause | Diagnostic Steps | Potential Solution |
|---|---|---|
| Insufficient drug exposure | Check pharmacokinetics (PK). Measure Cmax and AUC. Compare to efficacious levels in literature. | Reformulate for better solubility/bioavailability. Adjust dosing regimen (e.g., fractionated dosing). |
| Compensatory pro-angiogenic signaling | Analyze tumor lysates post-VDA for VEGF, SDF-1α, HIF-1α upregulation via ELISA/Western Blot. | Implement a scheduled combination with a targeted anti-angiogenic agent. |
| Innate vascular resistance | Perform pre-treatment vessel architecture analysis (immunofluorescence for α-SMA, pericyte coverage). | Pre-select models with immature vasculature or prime tumors with a VEGF inhibitor to destabilize mature vessels. |
Issue: Excessive Systemic Toxicity (e.g., Cardiotoxicity, Neuropathy) in Preclinical Models
| Possible Cause | Diagnostic Steps | Potential Solution |
|---|---|---|
| Off-target tubulin binding | Assess histopathology in heart and peripheral nerves. | Explore tumor-targeted liposomal or polymer-conjugated VDA formulations. |
| Cytokine storm | Monitor serum IL-6, TNF-α post-injection. | Implement a lower priming dose or pre-treat with anti-inflammatory agents (e.g., dexamethasone). |
| Exaggerated hemodynamic response | Monitor real-time blood pressure and ECG. | Switch to a slow intravenous infusion over bolus injection to blunt the acute response. |
Experimental Protocol: Assessing VDA Efficacy in a Heterogeneous Tumor Model
Objective: To evaluate the efficacy of a tubulin-binding VDA (e.g., Fosbretabulin/CA4P) while accounting for pre-existing vascular heterogeneity.
Materials:
- Orthotopic or subcutaneous tumor model (e.g., MDA-MB-231 breast carcinoma).
- VDA (CA4P, dissolved in saline).
- Small animal imaging system (MRI or ultrasound).
- MRI contrast agent (e.g., Gd-DTPA) or ultrasound microbubbles.
- Pimonidazole HCl (hypoxia marker).
- Fixation and immunohistochemistry reagents.
Procedure:
- Pre-treatment Stratification (Day -1):
- Anesthetize tumor-bearing animals.
- Acquire baseline DCE-MRI or CEUS images.
- Quantify tumor perfusion map. Segment tumors into High, Medium, and Low perfusion cohorts.
- Randomize animals within each perfusion cohort into Vehicle and VDA treatment groups.
VDA Administration (Day 0):
- Administer CA4P (e.g., 100 mg/kg, i.p.) or vehicle control at time T=0.
Acute Response Monitoring (Day 1):
- At T=4 hours post-dose, repeat perfusion imaging to quantify acute vascular shutdown.
Therapeutic Window Analysis (Day 1-3):
- Administer a secondary agent (e.g., chemotherapy, pimonidazole 60 mg/kg i.p.) at T=24 hours.
- For pimonidazole: Euthanize animals at T=30 hours. Harvest tumors, fix, and section for IHC staining of pimonidazole adducts and CD31.
Efficacy Endpoint (Day 7):
- Monitor tumor volumes daily.
- At study endpoint, perform terminal perfusion imaging and harvest tumors for histology (H&E, TUNEL, CD31).
Data Analysis:
- Correlate baseline perfusion parameters with the degree of acute vascular shutdown.
- Correlate acute shutdown with extent of necrosis and final tumor growth delay.
- Map the spatial relationship between residual perfusion (CD31+) and hypoxia (pimonidazole+) or secondary drug delivery.
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in VDA Research |
|---|---|
| Fosbretabulin (CA4P) | A leading tubulin-binding VDA prototype; disrupts endothelial cell cytoskeleton, causing rapid vascular shutdown. |
| Pimonidazole HCl | A hypoxia-activated marker; forms protein adducts in hypoxic regions (<10 mmHg O₂), used to identify the viable, perfused rim post-VDA. |
| DCE-MRI with Gd-DTPA | Gold-standard for perfusion quantification. Tracks contrast agent kinetics to derive quantitative parameters like Ktrans (transfer constant) and ve (extravascular extracellular space). |
| CD31/PECAM-1 Antibody | Standard immunohistochemical marker for vascular endothelial cells, used to quantify microvessel density and architecture. |
| α-Smooth Muscle Actin (α-SMA) Antibody | Marks pericytes and vascular smooth muscle cells; high coverage indicates mature, stabilized vessels which may be more resistant to VDA. |
| Recombinant VEGF / VEGF Trap (Aflibercept) | Used to manipulate the tumor vasculature—VEGF to prime, VEGF Trap to block compensatory signaling post-VDA. |
Visualizations
Title: VDA Mechanism, Heterogeneity Impact & Combination Strategies
Title: Experimental Workflow for VDA Studies in Heterogeneous Models
Computational Modeling of Tumor Hemodynamics for Predictive Delivery
Technical Support Center
Troubleshooting Guides & FAQs
Q1: Our computational fluid dynamics (CFD) simulation of drug transport consistently fails to converge when modeling highly permeable, chaotic vessel networks. What are the primary stability controls to adjust? A: Convergence failure in chaotic vasculature is often due to extreme permeability values and mesh distortion.
- Solution: Implement a dual-mesh adaptive approach. Use a coarse mesh for initial hemodynamic solution (velocity, pressure) and a refined, conforming mesh for the advection-diffusion-reaction (ADR) solute transport.
- Protocol:
- Run steady-state CFD on the coarse vascular mesh.
- Extract wall shear stress (WSS) data. Flag elements where WSS > 5 Pa or < 0.05 Pa.
- In these flagged regions, refine the mesh for the solute domain, ensuring a minimum of 5 elements across the vessel diameter.
- Map the hemodynamic solution onto the refined mesh.
- Run the transient ADR simulation with a reduced initial time step (e.g., 1e-4 s), using an implicit solver for stability.
Q2: When calibrating our model with in vivo imaging data (e.g., DCE-MRI), the predicted interstitial fluid pressure (IFP) gradient is significantly steeper than literature values. Which parameters are most sensitive? A: The interstitial hydraulic conductivity (K) and the lymphatic drainage coefficient (L) are the dominant sensitive parameters for IFP.
- Solution: Perform a local sensitivity analysis around your baseline values.
- Protocol:
- Define a baseline parameter set: K=3.5e-7 cm²/mmHg/s, L=1.1e-7 mmHg⁻¹s⁻¹.
- Vary each parameter by ±20% while holding others constant.
- Simulate and record the peak IFP and the IFP at the tumor rim.
- Calculate the normalized sensitivity coefficient:
S = (ΔOutput/Output_baseline) / (ΔParameter/Parameter_baseline).
Table 1: Sensitivity of Simulated IFP to Key Biophysical Parameters
| Parameter | Baseline Value | +20% Change in Parameter → %Δ in Peak IFP | Sensitivity Coefficient (S) |
|---|---|---|---|
| Vascular Permeability (P) | 2.5e-6 cm/s | +4.7% | 0.24 |
| Interstitial Hydraulic Conductivity (K) | 3.5e-7 cm²/mmHg/s | -18.2% | -0.91 |
| Lymphatic Drainage (L) | 1.1e-7 mmHg⁻¹s⁻¹ | -8.5% | -0.43 |
| Plasma Osmotic Pressure (π_c) | 20 mmHg | +1.1% | 0.06 |
Q3: How do we accurately define the boundary condition for drug influx from a leaky vessel in a discrete vasculature model? A: Use the Patlak equation (also known as the Kedem-Katchalsky flux) as a Neumann boundary condition at the vessel wall.
- Solution: The flux
J_sof solute across the vessel wall is:J_s = P * S * (C_p - C_i) + (1 - σ_f) * J_v * (C_p + C_i)/2, whereJ_vis the volumetric water flux from Starling's law. - Protocol for Implementation:
- From your CFD solution, extract
J_vand WSS at each vessel surface element. - Assign a local
Pvalue that is WSS-dependent (e.g.,P = P_base * (1 + α * log10(WSS/WSS_ref))). - For each time step in the transport simulation, calculate
J_susing the current plasma (C_p) and interstitial (C_i) concentrations. - Apply
J_sas a source term to the interstitial domain nodes adjacent to the vessel wall.
- From your CFD solution, extract
Q4: Our agent-based model (ABM) of cell response to a drug shows unrealistic, synchronized death. How can we introduce heterogeneity? A: This indicates missing intrinsic (genetic) and extrinsic (microenvironmental) variability.
- Solution: Parameterize each agent (cell) with a unique phenotype vector drawn from a multivariate distribution.
- Protocol:
- Define key phenotype parameters: drug uptake rate, metabolic rate, apoptosis threshold, and proliferation cycle length.
- For a population of N cells, define a mean vector (μ) and a covariance matrix (Σ) for these parameters based on experimental single-cell data.
- For each new agent, sample its parameter set from the multivariate normal distribution
N(μ, Σ). - Link the agent's local microenvironment (e.g., glucose concentration, IFP) to modulate these base parameters during the simulation.
Experimental Protocols for Model Validation
Protocol P1: In Vivo Measurement for Hemodynamic Parameter Calibration Objective: Acquire data to calibrate simulation boundary conditions and validate flow profiles. Materials: See Research Reagent Solutions below. Method:
- Animal Model: Implant tumor cells (e.g., MDA-MB-231 for breast cancer) orthotopically in nude mice (n=5).
- Vessel Labeling: Inject 100 µL of DyLight 488-labeled Lycopersicon esculentum (Tomato) Lectin (2 mg/mL in PBS) intravenously. This binds uniformly to endothelial glycocalyx.
- Imaging: After 3 minutes, euthanize the animal and excise the tumor. Image 1 mm thick slices using a high-resolution confocal microscope (e.g., Zeiss LSM 980) with a 20x objective. Acquire z-stacks (5 µm steps) of lectin-filled vasculature.
- Image Analysis: Use software (e.g., AngioTool, VesselVio) to skeletonize the network. Extract metrics: vessel diameter, length, tortuosity index, and branch point density per mm³.
- Data Integration: Use the diameter and branch point distribution to generate a synthetic, statistically equivalent vascular network for simulation inflow/outflow boundary setting.
Protocol P2: Ex Vivo Validation of Predicted Drug Distribution Objective: Compare computationally predicted drug distribution with actual ex vivo tissue measurements. Method:
- Simulation: Run your predictive delivery model for a specific nanoparticle (NP) (e.g., 100 nm PEGylated liposome, carrying a fluorescent dye). Export the 3D concentration map
C_sim(x,y,z,t)at t=1 hour post-administration. - In Vivo Administration: In a separate cohort of mice (n=5), administer the fluorescent NP intravenously at 10 mg/kg.
- Tissue Processing: At 1 hour post-injection, perfuse the mouse with cold PBS, excise the tumor, and snap-freeze in OCT compound. Section (10 µm thickness) using a cryostat.
- Quantification: Image sections using fluorescence microscopy. Co-stain with CD31 to identify blood vessels. Using image registration software (e.g., Elastix), align the experimental fluorescence image
I_expwith the corresponding slice fromC_sim. - Validation: Calculate the spatially resolved correlation coefficient (Pearson's R) between
I_expandC_simwithin the tumor region. A value of R > 0.7 indicates strong predictive validity.
Diagrams
Title: Research Workflow for Predictive Delivery
Title: Drug Transport Across Heterogeneous Vasculature
Research Reagent Solutions
Table 2: Essential Materials for Tumor Hemodynamics & Delivery Experiments
| Item | Function / Relevance | Example Product / Specification |
|---|---|---|
| Fluorescent Vascular Label | Labels perfused vasculature for 3D imaging and network analysis. Critical for defining model geometry. | DyLight 488 Lycopersicon esculentum Lectin (Vector Labs, DL-1174) |
| DCE-MRI Contrast Agent | Low molecular weight probe (e.g., Gd-DTPA) for in vivo measurement of vascular permeability (Ktrans) and perfusion. | Gadoterate meglumine (Dotarem) |
| Tunable Nanoparticles | Model drug carriers with controlled size (50-200 nm) and surface chemistry (PEG, targeting ligands) to validate size-dependent transport predictions. | PEGylated Liposomes (FormuMax, various sizes) |
| Pressure Transducer | Direct measurement of interstitial fluid pressure (IFP) for model validation via micropuncture. | Micropressure System (Model 5A, IPL-103, Instrumentation Laboratories) |
| Anti-CD31 Antibody | Immunohistochemical staining of endothelial cells for co-localization with drug signals in validation protocols. | Anti-CD31 (PECAM-1) Antibody (e.g., Abcam, ab28364) |
| Mathematical Solver Suite | Software for solving coupled CFD and advection-diffusion-reaction equations on complex geometries. | COMSOL Multiphysics with CFD and Chemical Reaction Modules |
Navigating Experimental Challenges: Optimizing Models and Protocols
This technical support center is designed to assist researchers in selecting and implementing preclinical models within the context of a broader thesis on Compensating for Tumor Vasculature Heterogeneity. Below are troubleshooting guides and FAQs addressing specific experimental challenges.
Frequently Asked Questions (FAQs)
Q1: My Patient-Derived Xenograft (PDX) model shows poor tumor take rates. What are the primary factors to consider? A: Poor engraftment can stem from insufficient tumor stromal support in the host. For vascular heterogeneity research, ensure the host mouse strain (e.g., NOD-scid IL2Rγnull [NSG]) is optimally immunosuppressed to support human vasculature cells. Implant tumor fragments (2-3 mm³) rather than single cells, and consider co-implantation of Matrigel to provide provisional stromal support. The site of implantation (orthotopic vs. subcutaneous) is also critical for maintaining native tumor vasculature signaling.
Q2: How do I validate that my orthotopic model accurately recapitulates the tumor microenvironment, especially the vasculature? A: Perform comparative histology and immunostaining on the original patient tumor and the orthotopic model. Key markers for vasculature heterogeneity include CD31 (pan-endothelial), MECA-32 (mouse-specific endothelial), and CD34 (human-specific endothelial). This allows you to quantify the degree of human vs. murine vessel contribution (a process called vascular mimicry or co-option) which is central to understanding heterogeneity.
Q3: My transgenic model does not respond to an anti-angiogenic drug that showed promise in cell lines. Is this expected? A: Yes. Transgenic models driven by a specific oncogene (e.g., PyMT for breast cancer) develop de novo tumors with a murine vasculature that may have evolved differently than human tumors. They lack the genetic heterogeneity of human cancers. This discrepancy underscores the need for models that incorporate human tumor stroma interactions. Consider validating in a PDX model where the human tumor vasculature is partially maintained or undergoes co-option.
Q4: What is the most significant technical challenge when switching from subcutaneous to orthotopic implants, and how can I mitigate it? A: The primary challenge is the surgical procedure and in vivo monitoring. Utilize in vivo imaging tools (e.g., luciferase-labeled tumor cells) for non-invasive tracking. For vasculature studies, ultrasound or photoacoustic imaging can monitor tumor blood flow and vessel density. Ensure rigorous post-operative care and use of analgesics to minimize stress, which can alter vascular perfusion and cytokine profiles.
Q5: How can I quantitatively compare vasculature heterogeneity across PDX, transgenic, and orthotopic models? A: Employ multiparametric analysis. Generate data as summarized in the table below, combining vessel density, pericyte coverage, hypoxia markers, and species-specific endothelial cell quantification.
| Model Parameter | Patient-Derived Xenograft (PDX) | Transgenic (e.g., PyMT) | Orthotopic (Cell-Line Derived) |
|---|---|---|---|
| Tumor Take Rate (%) | 20-70 (highly sample-dependent) | 100 (by design) | 80-95 |
| Time to Tumor Onset | 2-8 months (slow, variable) | 6-12 weeks (predictable) | 2-4 weeks (fast) |
| Genetic Heterogeneity | High (reflects patient tumor) | Low (driver oncogene + acquired mutations) | Moderate (clonal cell line) |
| Stromal/Vasculature Origin | Mixed human (initial) & murine (host-derived) | 100% murine | 100% murine |
| Key Advantage for Vascular Studies | Retains human tumor vasculature signaling for 1-2 passages; studies of vessel co-option | Studies of de novo tumorigenesis & angiogenesis in immune-competent host | Organ-specific vascular microenvironment influences metastasis |
| Major Limitation for Vascular Studies | Gradual loss of human stromal cells; expensive; time-consuming | May not mimic human vascular signaling pathways | Vasculature originates from a homogeneous cell line, not a heterogeneous tumor |
Experimental Protocols
Protocol 1: Establishing a PDX Model for Vascular Heterogeneity Analysis
- Tumor Processing: Under sterile conditions, mince a fresh patient tumor sample (chemo-naïve preferred) in cold PBS to ~2-3 mm³ fragments.
- Implantation: Use a 10-gauge trocar to implant one fragment subcutaneously into the flank of an 8-week-old NSG mouse. For orthotopic PDX, implant into the corresponding organ capsule (e.g., mammary fat pad).
- Monitoring: Measure tumors twice weekly. Upon reaching 1000-1500 mm³, harvest. Pass serially by re-implanting fragments into new mice (P1, P2, etc.).
- Vascular Analysis: At each passage, analyze one fragment by IHC for human (CD34) and mouse (MECA-32) endothelial markers to track stromal evolution.
Protocol 2: Assessing Vessel Maturity and Perfusion in an Orthotopic Model
- Labeling: Inject mice bearing orthotopic tumors with:
- Pimonidazole (60 mg/kg, i.p.) 1 hour before sacrifice to label hypoxic regions.
- FITC-labeled Lycopersicon esculentum (Tomato) Lectin (100 µL of 1 mg/mL, i.v.) or Dye-labeled anti-CD31 antibody 5 minutes before sacrifice to label perfused vessels.
- Tumor Harvest: Excise tumor, freeze in O.C.T. compound.
- Immunofluorescence: Section (10 µm) and stain for CD31 (vessels), α-SMA (pericytes), and pimonidazole adducts.
- Quantification: Use image analysis software (e.g., ImageJ) to calculate: % lectin+CD31+ vessels (perfused fraction), and % CD31+ vessels with α-SMA+ coverage (mature fraction). Correlate with hypoxic areas.
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in Vasculature Heterogeneity Research |
|---|---|
| NSG (NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ) Mice | Immunodeficient host for PDX, allows engraftment of human tumor and stromal elements with minimal rejection. |
| Growth Factor-Reduced Matrigel | Basement membrane extract. Co-injected with tumor cells to provide provisional stromal support and enhance tumor take, influencing early angiogenesis. |
| Species-Specific Antibodies (Anti-human CD34, Anti-mouse MECA-32) | Critical for distinguishing human-derived vs. mouse-derived endothelial cells in PDX models via IHC/IF. |
| Pimonidazole HCl | Hypoxia probe. Forms protein adducts in cells with pO₂ < 10 mm Hg, allowing mapping of poorly vascularized regions. |
| FITC-Lectin (L. esculentum) | Vascular perfusion marker. Binds to endothelial glycocalyx only in functional, perfused blood vessels when injected intravenously. |
| In Vivo Imaging System (IVIS) with Luciferin | Enables non-invasive, longitudinal tracking of tumor growth and metastatic spread in orthotopic and transgenic models. |
Visualizations
Title: Decision Flow for Choosing Preclinical Model
Title: Workflow for Tumor Vasculature Analysis
Technical Support Center: Troubleshooting Guides & FAQs
FAQ 1: Why do I observe high inter-animal variability in perfusion metrics (e.g., Ktrans) within my murine tumor model, even with standardized inoculation?
Answer: High variability often stems from unaccounted-for intrinsic tumor vasculature heterogeneity, a core focus of our broader thesis research. Key pitfalls include:
- Tumor Region Selection: Measurements taken from a single, central ROI miss peripheral vascular hot spots or necrotic cores. Solution: Implement multi-ROI or whole-tumor histogram analysis.
- Anesthetic & Physiological State: Isoflurane and ketamine/xylazine differentially impact cardiac output and blood pressure, altering perfusion. Solution: Standardize anesthetic protocol, monitor body temperature, and consider physiological gating.
- Contrast Agent Kinetics Model Misapplication: Using the Tofts model for tumors with severe plasma volume fraction (vp) or permeability can bias Ktrans. Solution: Validate with the Extended Tofts or Patlak model. See Table 1.
FAQ 2: My dynamic contrast-enhanced (DCE) MRI data shows poor signal-to-noise ratio (SNR), leading to unreliable permeability parameter fitting. How can I improve this?
Answer: Poor SNR compromises the arterial input function (AIF) and tissue curve fidelity.
- Primary Cause - Insufficient Temporal vs. Spatial Resolution Trade-off: Too-fast sampling reduces SNR per time point; too-slow sampling misses AIF peak.
- Troubleshooting Protocol:
- Pre-scan: Determine the optimal trade-off. For typical tumor studies, a temporal resolution of 5-15 seconds is often viable.
- Contrast Administration: Ensure rapid, bolus injection (<5s) via a tail vein catheter, followed by a saline flush. Slow injection smears the AIF.
- Data Processing: Apply appropriate noise-reduction filters (e.g., Gaussian spatial smoothing) before pharmacokinetic modeling, not after.
FAQ 3: In longitudinal studies, how do I distinguish true anti-angiogenic drug effect from day-to-day measurement variability in vessel permeability?
Answer: Distinguishing effect from noise requires rigorous controls and standardized analysis.
- Implement Baseline Stratification: Randomize treatment groups based on Day 0 perfusion metrics to ensure equivalent starting heterogeneity.
- Control for Scan-to-Scan Variability: Include a cohort of untreated control tumors scanned longitudinally. The standard deviation in their metrics defines the "noise floor" for detecting a true effect.
- Use Paired Statistical Tests: Compare each tumor to its own baseline using paired tests (e.g., paired t-test, Wilcoxon signed-rank), which are more sensitive to longitudinal change.
FAQ 4: When using laser speckle contrast imaging (LSCI) for surface perfusion, how do I correct for motion artifacts and underlying vessel geometry interference?
Answer: LSCI is highly sensitive to movement and large static vessels.
- Motion Artifact Protocol:
- Rigidly immobilize the animal and tumor window chamber.
- Apply digital image stabilization algorithms post-hoc.
- Use a region-growing algorithm to exclude areas with sudden, large spatial discontinuities in flow.
- Vessel Geometry Protocol: Large vessels show artificially high speckle contrast. Apply a vessel segmentation algorithm (e.g., Frangi filter) to mask out major vessels from parenchymal perfusion analysis.
Data Presentation
Table 1: Common Pharmacokinetic Models for DCE-MRI Analysis: Applications and Pitfalls
| Model | Key Parameters | Best For | Major Pitfall in Heterogeneous Tumors |
|---|---|---|---|
| Tofts (Standard) | Ktrans, ve | Low permeability surfaces (Ktrans << Fp, plasma flow) | Overestimates Ktrans if vp > ~10%; ignores plasma volume. |
| Extended Tofts | Ktrans, ve, vp | Most solid tumors; accounts for plasma volume. | Can be unstable if AIF is poorly defined or data is noisy. |
| Patlak | Ktrans, vp | High permeability surfaces (PS >> Fp) where backflux is negligible early on. | Only valid for initial ~2-3 minutes; underestimates if permeability is low. |
| 2-Compartment Exchange (2CXM) | Fp, PS, vp, ve | Separates flow (Fp) from permeability-surface area product (PS). | Requires very high-temporal-resolution, high-SNR data; complex fitting. |
Table 2: Impact of Anesthetic on Murine Cardiovascular Physiology & Perfusion Metrics
| Anesthetic Regimen | Mean Arterial Pressure (Change) | Heart Rate (Change) | Cardiac Output (Change) | Recommended for Perfusion Studies? |
|---|---|---|---|---|
| Isoflurane (1-2% in O₂) | ↓↓ (Severe decrease) | ↓/↑ (Variable) | ↓↓ | Use with caution; must monitor & maintain temperature. |
| Ketamine/Xylazine (i.p.) | ↓ (Moderate decrease) | ↓↓ (Bradycardia) | ↓ | Longer, stable plane; better for terminal studies. |
| Medetomidine/Fentanyl/ Midazolam (i.p.) | ↓ (Initial) then stable | ↓ (Stable) | ↓ (Stable) | Provides stable physiology; requires reversal agent. |
| Awake, Restrained | Baseline (Normal) | Baseline (Normal) | Baseline (Normal) | Ideal for physiology; high stress can confound results. |
Experimental Protocols
Protocol 1: Robust Multi-Parametric DCE-MRI in Subcutaneous Tumor Models Objective: To quantify tumor perfusion (Ktrans, Fp) and interstitial volume (ve) while compensating for heterogeneity.
- Animal Preparation: Anesthetize mouse using a pre-optimized, consistent protocol (e.g., medetomidine cocktail). Maintain core temperature at 37°C using a warm air system. Place a 27G tail vein catheter for contrast delivery.
- MRI Acquisition: Use a high-field (≥7T) scanner. Acquire high-resolution T1 maps. For DCE, use a fast T1-weighted gradient echo sequence (TR/TE ~5/2 ms, flip angle 15-25°). Temporal resolution: 10 seconds per volume for 15-20 minutes.
- Contrast Injection: At the 6th dynamic time point, inject Gadobutrol (0.1 mmol/kg) at 10 mL/min via syringe pump, followed by 0.3 mL saline flush.
- Data Analysis:
- Define AIF from the iliac artery or heart ventricle using a population-based AIF corrected for individual hematocrit.
- Segment tumor using semi-automated thresholding. Analyze whole tumor, then sub-divide into core, rim, and total via histogram analysis.
- Fit time-concentration curves on a voxel-by-voxel basis using the Extended Tofts model. Report median, 75th percentile, and heterogeneity index (75th/25th percentile ratio) for Ktrans.
Protocol 2: Correlative LSCI and Histology for Validation of Vascular Heterogeneity Objective: To validate non-invasive perfusion maps with ex vivo microvascular morphology.
- In Vivo LSCI: Under terminal anesthesia, expose the tumor surface. Acquire speckle images at 10 Hz for 2 minutes using a 785 nm laser. Calculate speckle contrast (K2) and convert to relative blood flow units using a pseudo-colour scale.
- Tissue Processing & Staining: Immediately euthanize and excise tumor. Slice along the LSCI imaging plane. Fix in 4% PFA.
- Immunofluorescence: Section (5 µm). Stain for CD31 (endothelial cells, Alexa Fluor 488), α-SMA (pericytes, Cy3), and Hoechst (nuclei). Image using confocal microscopy.
- Histology: Adjacent section. Stain with H&E for necrosis assessment.
- Image Registration & Analysis: Register in vivo LSCI flow map to ex vivo whole-slide scans using landmark-based registration. Correlate regional flow values with corresponding regional microvessel density (MVD), pericyte coverage index (% CD31+ vessels co-stained with α-SMA), and necrotic fraction.
Mandatory Visualization
The Scientist's Toolkit: Research Reagent Solutions
| Item | Category | Function & Relevance to Standardization |
|---|---|---|
| Gadobutrol (Gadovist) | MRI Contrast Agent | High relaxivity (r1) gadolinium-based agent. Provides stronger signal per unit concentration for more precise DCE-MRI pharmacokinetic modeling. |
| Dextran-Texas Red (70 kDa) | Fluorescent Vascular Tracer | Used in intravital microscopy to measure vascular permeability. Large molecular weight approximates macromolecular leakage. Quantifies extravasation rate. |
| Anti-CD31 Antibody | Immunohistochemistry Reagent | Labels endothelial cells for microvessel density (MVD) calculation, a gold-standard histology correlate for perfusion maps. |
| Anti-αSMA Antibody | Immunohistochemistry Reagent | Labels pericytes and smooth muscle cells. Used to calculate pericyte coverage index, a key marker of vessel maturity and stability affecting permeability. |
| Hoechest 33342 | Nuclear Stain / Perfusion Tracer | When injected in vivo, labels perfused vessels (via diffusion into endothelial nuclei). Correlates immediate perfusion with vascular architecture post-sectioning. |
| Medetomidine/Ketamine Cocktail | Anesthetic | Provides prolonged, stable anesthetic plane with relatively preserved cardiovascular function compared to inhalants, reducing a major source of perfusion variability. |
| Physiological Monitoring System | Equipment | Measures core temperature, respiration rate, and/or ECG. Critical for maintaining and reporting consistent physiological conditions during in vivo imaging. |
| Population-Based AIF Package | Software/Algorithm | Mitigates errors from individual AIF measurement. Uses a pre-characterized, species/strain-specific AIF shape, scaled by individual hematocrit or cardiac output. |
Optimizing Dosing Schedules for Combination Therapies (e.g., Anti-angiogenics + Chemo)
Troubleshooting Guide & FAQ
This Technical Support Center is framed within the research thesis "Compensating for Tumor Vasculature Heterogeneity to Improve Therapeutic Delivery and Efficacy." The following Q&A addresses common experimental challenges when optimizing combination therapy schedules.
Q1: In our mouse xenograft model, administering an anti-angiogenic agent (e.g., bevacizumab) before chemotherapy (e.g., paclitaxel) reduces tumor size initially, but long-term control is worse than concurrent scheduling. What might be happening?
A: This likely relates to vascular normalization. Premature or excessive anti-angiogenic pruning can lead to vascular collapse, increasing hypoxia and interstitial fluid pressure. This creates a physical barrier to subsequent chemotherapeutic drug delivery. The "window of normalization"—a temporary period of improved vessel structure and function—is likely missed. Troubleshooting steps:
- Monitor the Window: Use dynamic contrast-enhanced MRI (DCE-MRI) or intravital microscopy to assess perfusion and vascular permeability at multiple time points (e.g., 1, 2, 4, 6 days) post anti-angiogenic dose.
- Schedule Chemotherapy at Peak Normalization: Time chemotherapy administration to coincide with observed peak perfusion and reduced hypoxia (e.g., day 2-4 post anti-angiogenic dose in many models).
- Titrate Dose: A lower dose of the anti-angiogenic agent may induce normalization without causing excessive regression.
Q2: Our data on tumor perfusion after combination therapy is highly variable between subjects, making schedule optimization difficult. How can we account for this?
A: High variability is a direct manifestation of tumor vasculature heterogeneity. This inter- and intra-tumor heterogeneity in vessel density, maturity, and pericyte coverage leads to non-uniform drug delivery.
- Stratify by Biomarkers: Pre-treatment, segment tumors using imaging biomarkers (e.g., baseline Ktrans from DCE-MRI, or HIF-1α staining). Design arms that schedule chemotherapy based on these sub-populations.
- Use a Feedback-Driven Adaptive Schedule: Instead of a fixed schedule, design an experiment where the timing of the subsequent chemotherapy dose is determined by a real-time perfusion readout (e.g., ultrasound-guided photoacoustics for hemoglobin oxygen saturation).
- Incorporate Stromal-Targeting Agents: Consider adding a stroma-modulating agent (e.g., PEGPH20) to a sub-arm to homogenize the tumor microenvironment and reduce delivery heterogeneity, then test the combination schedule.
Q3: When testing different dosing sequences in vitro, how can we best model the impact of vascular changes on chemo delivery?
A: Standard 2D co-cultures fail to capture vascular dynamics. Implement a 3D microfluidic model.
- Protocol: Modeling Vasculature-Chemo Delivery in a Chip:
- Seed endothelial cells (e.g., HUVECs) in the central channel of a collagen I/Matrigel mixture in a microfluidic device. Perfuse with medium containing angiogenic factors (VEGF) to form a microvascular network over 5-7 days.
- Seed tumor spheroids in adjacent chambers.
- Pre-treatment Phase: Perfuse the vascular channel with the anti-angiogenic agent (e.g., a VEGFR2 inhibitor like sunitinib) at clinically relevant concentrations for 48 hours.
- Treatment Phase: At designated times (24h, 72h, 120h) post anti-angiogenic initiation, add a fluorescently tagged chemotherapy agent (e.g., doxorubicin) to the perfusion medium.
- Quantification: Use live-cell imaging to track fluorescence intensity within the tumor spheroid over time to generate a pharmacokinetic profile for each scheduling regimen.
Q4: What are the key molecular markers to assess the vascular normalization phenotype in tissue samples?
A: Analyze a panel of markers, as no single marker is definitive. Prioritize spatial analysis (multiplex IHC) over bulk analysis.
| Marker Category | Specific Marker | Interpretation in Normalization |
|---|---|---|
| Pericyte Coverage | α-SMA, NG2, Desmin | Increased association with CD31+ vessels. |
| Basement Membrane | Collagen IV | Thickened and more continuous. |
| Vessel Maturity | PECAM-1 (CD31) / Endomucin Ratio | Higher ratio indicates maturity. |
| Oxygenation/Hypoxia | HIF-1α, CAIX | Reduced expression. |
| Proliferation | Ki67 in endothelial cells | Reduced endothelial cell proliferation. |
Experimental Protocols
Protocol 1: Determining the Vascular Normalization Window In Vivo Objective: To identify the optimal time window for chemotherapy delivery following anti-angiogenic therapy. Materials: Syngeneic or xenograft tumor model, Anti-angiogenic agent (e.g., Bevacizumab, 10 mg/kg), Chemotherapy agent (e.g., Cisplatin, 3 mg/kg), Lectin-FITC (for perfusion), Pimonidazole (for hypoxia). Method:
- Randomize tumor-bearing animals into cohorts (n≥5).
- Administer a single dose of anti-angiogenic agent or vehicle (Day 0).
- On Days 1, 2, 3, 4, and 7 post-treatment, inject one cohort per time point with Lectin-FITC (intravenous, 5 minutes before sacrifice) and Pimonidazole (intraperitoneal, 1 hour before sacrifice).
- Harvest tumors, snap-freeze, and prepare sections.
- Perform immunofluorescence staining for CD31 (vessels), analyze Lectin-FITC (perfused vessels), and pimonidazole adducts (hypoxic regions).
- Quantitative Analysis: Calculate perfused vessel fraction (Lectin+CD31+/Total CD31+ area) and hypoxic fraction (Pimo+ area / Total area). The peak perfused fraction coinciding with reduced hypoxic fraction defines the normalization window.
Protocol 2: Adaptive Scheduling Based on Real-Time Perfusion Imaging Objective: To implement a feedback-controlled dosing schedule for chemotherapy. Materials: Tumor model with dorsal window chamber or amenable to high-frequency ultrasound, Anti-angiogenic agent, Chemotherapy agent, Ultrasound/Photoacoustic system. Method:
- Implant tumors and allow growth to ~100 mm³.
- Administer the first dose of anti-angiogenic agent.
- Daily Monitoring: Acquire non-invasive photoacoustic images to quantify tumor hemoglobin oxygen saturation (sO₂), a surrogate for perfusion and oxygenation.
- Decision Point: Set a threshold (e.g., 15% increase from baseline sO₂). When an individual subject's tumor sO₂ reaches this threshold, administer the scheduled dose of chemotherapy.
- Compare outcomes (tumor growth delay, survival) against cohorts on fixed calendar schedules.
Signaling Pathways in Vascular Normalization
Title: Anti-VEGF Signaling & Vascular Normalization Pathways
Experimental Workflow for Schedule Optimization
Title: Workflow for Testing Dosing Schedules In Vivo
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function & Application in Schedule Optimization |
|---|---|
| Recombinant Anti-VEGF (Bevacizumab analog) | Induces vascular normalization in vivo. Used to establish the priming window for chemotherapy. |
| Lectin-FITC (e.g., Lycopersicon Esculentum) | Labels perfused blood vessels when injected intravenously shortly before sacrifice. Critical for quantifying functional vasculature. |
| Hypoxia Probes (Pimonidazole HCl) | Forms protein adducts in hypoxic regions (<10 mmHg O₂). Immunodetection allows mapping of tumor hypoxia dynamics post-treatment. |
| CD31/PECAM-1 Antibody | Standard marker for pan-endothelial cell staining to quantify total vessel density. |
| α-Smooth Muscle Actin (α-SMA) Antibody | Marks pericytes and vascular smooth muscle cells. Co-staining with CD31 assesses vessel maturity/pericyte coverage. |
| Matrigel Basement Membrane Matrix | Used for 3D endothelial cell tube formation assays in vitro and for enriching tumor implants in vivo. |
| Microfluidic Co-culture Devices (e.g., from AIM Biotech) | Enables creation of 3D, perfusable vascular networks alongside tumor cells to model drug delivery kinetics. |
| Dextran-Texas Red (70 kDa) | Fluorescent vascular tracer to assess vascular permeability in vivo or in microfluidic devices. |
Addressing Inter- and Intra-tumor Heterogeneity in Study Design
Technical Support Center: Troubleshooting Guides & FAQs
Frequently Asked Questions
Q1: Our preclinical drug efficacy results are highly variable between mice, even within the same treatment group. What could be the cause, and how can we mitigate this? A: This is a classic symptom of unaccounted intra-tumor variability, often driven by heterogeneous vasculature leading to inconsistent drug delivery. Mitigation strategies include:
- Increase Sample Size: Power your studies to detect effects despite higher variance.
- Implement Stratified Randomization: Prior to treatment, stratify animals based on tumor volume, vascular density (via pre-imaging), or biomarker expression.
- Use Orthotopic Models: Subcutaneous models often have less complex vasculature. Orthotopic models better recapitulate the native tumor microenvironment and its vascular heterogeneity.
- Pharmacodynamic (PD) Biomarkers: Measure drug target engagement or pathway modulation in addition to tumor volume.
Q2: When sampling a tumor for biomarker analysis, which region should we biopsy to get a representative result? A: A single biopsy is rarely representative due to intra-tumor heterogeneity. The recommended protocol is:
- Multi-Region Sampling: If possible, take multiple cores from the same tumor, annotating location (e.g., core, periphery, necrotic edge).
- Image-Guided Biopsy: Use companion diagnostic imaging (e.g., DCE-MRI for high-perfusion areas, PET for metabolically active regions) to guide sampling toward regions of biological interest.
- Clearly Document: Any single biopsy result must be reported with the caveat of its spatial context within the tumor.
Q3: Our imaging data (DCE-MRI) shows extreme variability in vascular perfusion parameters (Ktrans, ve) across tumors in the same cohort. How should we analyze this data? A: Do not just average parameters across the whole tumor. Heterogeneity is the data.
- Voxel-Based Analysis: Analyze parameter distributions (histograms) for each tumor.
- Habitat Mapping: Segment the tumor into distinct vascular "habitats" (e.g., high-Ktrans rim, low-Ktrans core). Quantify the volume or spatial arrangement of each habitat.
- Use Heterogeneity Metrics: Calculate statistical measures of dispersion (e.g., standard deviation, interquartile range, entropy) of perfusion parameters as biomarkers themselves.
Q4: How many patient-derived xenograft (PDX) lines or cell line-derived models are needed to account for inter-tumor variability in therapy screening? A: Using a single model gives misleading results. The table below summarizes recommendations based on recent consensus literature.
Table 1: Model Selection Recommendations for Accounting for Inter-Tumor Variability
| Study Phase | Minimum Number of Distinct Models | Rationale & Criteria for Selection |
|---|---|---|
| Initial Screening | 3-5 | Should represent distinct molecular subtypes (e.g., basal vs. luminal) or genetic backgrounds (e.g., KRAS mutant vs. WT). |
| Lead Optimization | 5-8 | Include models with known/resistant to standard of care, and varying levels of vascular maturity. |
| Preclinical Efficacy | 8-15+ | Use a panel that reflects the clinical prevalence of subtypes. PDX models are strongly preferred at this stage. |
Experimental Protocols
Protocol 1: Multi-Region Tumor Sampling for Genomic and Histological Analysis Objective: To capture intra-tumor heterogeneity from a resection specimen. Materials: Fresh tumor specimen, sterile surgical blades, RNAlater, 10% Neutral Buffered Formalin, cryomold with O.C.T. compound, dry ice, liquid nitrogen. Procedure:
- Immediately after resection, photograph and orient the specimen.
- Section the tumor transversely into 3-5 mm slices.
- From each slice, use a biopsy punch or blade to sample from at least three distinct regions: the invasive margin, the tumor core, and an intermediate area. Avoid grossly necrotic tissue.
- Divide each sample tripartitely: (a) Snap-freeze in O.C.T. on dry ice for cryosectioning; (b) Place in RNAlater for genomics/transcriptomics; (c) Fix in formalin for 24-48hrs for FFPE blocks.
- Clearly label all samples with a unique Tumor ID and Region Code (e.g., T1Core, T1Margin).
Protocol 2: Dynamic Contrast-Enhanced (DCE) MRI for Quantifying Vascular Heterogeneity Objective: To non-invasively map spatial variations in tumor perfusion and permeability. Materials: Small animal MRI, physiological monitoring equipment, tail vein catheter, gadolinium-based contrast agent (e.g., Gd-DTPA), analysis software (e.g., MITK, OsiriX). Procedure:
- Anesthetize and position the tumor-bearing mouse. Place a tail vein catheter.
- Acquire baseline T1 maps using a variable flip angle sequence.
- Initiate a fast T1-weighted gradient echo sequence. After ~5 baseline images, automatically inject contrast agent (0.1-0.2 mmol/kg) via catheter.
- Continue imaging for 15-30 minutes post-injection to capture washout kinetics.
- Analyze data using a pharmacokinetic model (e.g., Tofts model) on a voxel-by-voxel basis to generate parameter maps for Ktrans (transfer constant), ve (extravascular extracellular space), and kep (rate constant).
- Use histogram analysis or clustering algorithms on parameter maps to quantify heterogeneity.
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for Tumor Vasculature Studies
| Reagent / Material | Function & Application |
|---|---|
| CD31/PECAM-1 Antibody | Immunohistochemistry marker for pan-endothelial cells, used to quantify microvessel density (MVD). |
| α-SMA Antibody | Marks pericytes and vascular smooth muscle cells; assesses vessel maturity (pericyte coverage). |
| HIF-1α Antibody | Immunofluorescence marker for hypoxic regions, often inversely correlated with vasculature. |
| Pimonidazole HCl | Hypoxia probe. Injected in vivo, forms adducts in hypoxic (<1.3% O2) regions, detectable by IHC. |
| Dextran-Texas Red (70 kDa) | Fluorescent vascular permeability tracer. Injected IV, its extravasation indicates vessel leakiness. |
| Matrigel (Growth Factor Reduced) | Used for in vitro endothelial tube formation assays and for implanting tumor cells in vivo. |
| VEGFR2 (Kinase Insert Domain Receptor) Inhibitor (e.g., SU5416) | Pharmacologic tool to disrupt angiogenic signaling in validation experiments. |
Diagrams
Diagram 1: Key Pathways in Tumor Vasculature Heterogeneity
Diagram 2: Multi-Region Profiling Experimental Workflow
Best Practices for In Vivo Imaging of Tumor Vasculature (MRI, Photoacoustics, IVM)
Technical Support Center: Troubleshooting & FAQs
This support center is designed to assist researchers integrating multi-modal imaging to characterize tumor vasculature heterogeneity, a core challenge in developing effective vascular-targeting therapies. The guidance is framed within the thesis context: "Compensating for tumor vasculature heterogeneity requires robust, multi-parametric in vivo imaging to validate predictive models and assess treatment modulation."
FAQ & Troubleshooting Guide
Q1: In Dynamic Contrast-Enhanced MRI (DCE-MRI), my kinetic modeling (e.g., Tofts model) yields highly variable Ktrans values within the same tumor, sometimes with unrealistic negatives. What are the primary sources of error? A: This directly reflects the challenge of quantifying heterogeneous perfusion. Key issues are:
- AIF Selection: The Arterial Input Function (AIF) is critical. A population-based AIF may not reflect individual physiology.
- Fix: Implement an image-derived AIF from a major artery (e.g., caudal artery in mice) within the same scan. Ensure high temporal resolution (~1-3 sec) for the initial phase.
- Motion Artifact: Respiratory motion blurs tissue boundaries.
- Fix: Use physiological gating (respiratory) during acquisition or apply robust image registration during post-processing.
- Low Temporal vs. Spatial Resolution Trade-off: High spatial resolution for heterogeneity analysis often compromises temporal resolution.
- Fix: Prioritize temporal resolution for accurate kinetic modeling; use a larger voxel size if necessary.
Q2: During Photoacoustic Imaging, my vascular oxygen saturation (sO₂) maps appear noisier in deeper tumor regions. How can I improve signal quality? A: This is due to light scattering and attenuation.
- Spectral Unmixing Error: Insufficient wavelength sampling or dominance of hemoglobin signals.
- Fix: Increase the number of wavelengths sampled (≥10 across 680-900 nm). Confirm the absence of other absorbing chromophores (e.g., reporter dyes) that could confound unmixing.
- Light Delivery: Non-uniform illumination.
- Fix: Use a diffuse light delivery system or rotate the animal/imager to provide multi-directional illumination and improve depth penetration.
Q3: For Intravital Microscopy (IVM), I struggle with maintaining tumor vessel visibility over long-term (longitudinal) imaging sessions due to window clouding or tissue growth. A: This is a common hurdle for longitudinal heterogeneity studies.
- Chronic Window Clouding: Inflammation and fibrosis.
- Fix: Use biocompatible, gas-permeable titanium window setups. Administer anti-inflammatory drugs (e.g., dexamethasone) peri-operatively and in imaging maintenance doses, as approved by animal protocol.
- Vessel Labeling Fade: Photobleaching or antibody clearance.
- Fix: For fluorescent albumin or lectin for perfused vasculature, re-inject prior to each imaging session. Consider using transgenic animal models with fluorescently tagged endothelial markers (e.g., Tie2-GFP).
Q4: How do I best co-register data between high-resolution IVM and whole-tumor MRI/Photoacoustics to validate heterogeneity maps? A:
- Strategy: Use fiducial markers visible across modalities (e.g., small, inert silicone beads) implanted at the tumor periphery during window chamber installation.
- Protocol: Acquire MRI/PA image first. Before IVM, acquire a low-magnitude, large field-of-view "scout" image of the window chamber and tumor with fiducials. Use the fiducials and the tumor outline as landmarks for manual or semi-automated 3D registration in software like 3D Slicer or MATLAB.
Experimental Protocols
Protocol 1: Multi-Parametric MRI for Vascular Heterogeneity
- Animal Model: Orthotopic or dorsal window chamber tumor (e.g., 4T1, CT26).
- Anesthesia: Use 1-2% isoflurane with medical air/oxygen. Maintain body temperature.
- Contrast Agent: Gadoterate meglumine (Gd-DOTA), 0.1-0.2 mmol/kg, injected via tail vein cannula.
- MRI Acquisition (9.4T preclinical system):
- Localizer: Fast gradient echo.
- T1 Mapping: Use variable flip angle method (e.g., 2°, 10°, 20°).
- DCE-MRI: Run a T1-weighted fast spoiled gradient echo sequence. Temporal resolution: 3 sec. Total duration: 10-15 minutes. Inject contrast at frame 5.
- Anatomical Scan: High-resolution T2-weighted RARE sequence for tumor morphology.
- Analysis: Use software (e.g., MITK, Horos) to draw ROI. Calculate AIF, apply extended Tofts model to generate parametric maps (Ktrans, ve).
Protocol 2: Multi-Spectral Optoacoustic Tomography (MSOT) for sO₂
- Animal Preparation: Depilate tumor region. Apply ultrasound coupling gel.
- Anesthesia: Induce with 3% isoflurane, maintain at 1.5-2%.
- Positioning: Place animal in prone position in the MSOT holder with membrane for coupling.
- Image Acquisition:
- Acquire a baseline scan at multiple wavelengths (e.g., 700, 730, 760, 800, 850 nm).
- Optionally, administer intravascular agent (e.g., Angiosense 680, 2 nmol IV) for enhanced vascular contrast.
- Acquire post-injection multi-spectral data set.
- Analysis: Use built-in linear unmixing or spectral fitting algorithm to separate oxy- and deoxy-hemoglobin signals. Calculate sO₂ = [HbO₂] / ([HbO₂] + [Hb]).
Quantitative Data Summary: Imaging Modalities Comparison
| Parameter | MRI (DCE) | Photoacoustics (MSOT) | Intravital Microscopy (IVM) |
|---|---|---|---|
| Resolution | 50-100 µm | 50-150 µm | 1-5 µm |
| Penetration Depth | Unlimited (whole body) | 5-10 mm | < 500 µm |
| Key Vascular Metrics | Ktrans, ve, Blood Flow | sO₂, Total Hemoglobin, Vessel Density | Vessel Diameter, Permeability, RBC Velocity, Leukocyte Rolling |
| Temporal Resolution | Seconds-Minutes | Seconds | Milliseconds-Seconds |
| Throughput | Moderate (1-2 animals/hr) | High (10+ animals/hr) | Low (long-term setup per animal) |
| Primary Heterogeneity Data | Macro-regional perfusion & permeability | Hemodynamic oxygen gradients | Single-vessel dynamic behavior |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Application |
|---|---|
| Gd-based Contrast Agent (e.g., Gd-DOTA) | Small molecular weight MRI contrast agent for DCE-MRI, extravasates in leaky vasculature, informing on permeability (Ktrans). |
| Fluorescent Dextrans (e.g., 70 kDa FITC-Dextran) | IVM reagent for labeling the plasma volume. Used to quantify vascular permeability and blood flow dynamics. |
| Anti-CD31 Antibody (Fluorescent conjugate) | IVM reagent for pan-endothelial labeling to visualize total vascular network architecture. |
| Angiosense 680/750 | Long-circulating vascular-labeled optoacoustic/Fluorescent agent for enhancing PA and IVM signal from blood pool. |
| Hoechst 33342 | DNA-binding dye for IVM; when injected IV, labels perfused nuclei, aiding in defining perfused vs. total vasculature. |
| Isoflurane | Volatile inhalation anesthetic for stable, long-term maintenance of animal physiology during imaging sessions. |
| Physiological Monitoring System | Monitors and maintains core body temperature, respiration, and heart rate, essential for reproducible hemodynamic data. |
Visualization Diagrams
Title: Multimodal Imaging Workflow for Vascular Heterogeneity
Title: DCE-MRI Contrast Kinetics & Tofts Model
From Bench to Bedside: Validating Efficacy and Comparing Therapeutic Platforms
Technical Support Center: Troubleshooting Guide & FAQs
Welcome to the technical support center for research on Compensating for Tumor Vasculature Heterogeneity. This guide addresses common experimental issues with three primary delivery platforms.
FAQs & Troubleshooting
Nanoparticles (e.g., Liposomes, Polymeric NPs)
Q1: My nanoparticle formulation shows low drug encapsulation efficiency (EE). How can I improve it? A: Low EE is often due to drug-polymer/lipid mismatch or suboptimal preparation method.
- Troubleshooting: For hydrophobic drugs in polymeric NPs, use nanoprecipitation with a water-miscible organic solvent (e.g., acetone). For hydrophilic drugs in liposomes, employ the ammonium sulfate gradient or remote loading method. Increasing the drug-to-carrier ratio during synthesis may also help, but monitor for stability issues.
Q2: My targeted nanoparticles exhibit high non-specific uptake in the liver and spleen, reducing tumor accumulation. A: This is a common issue related to opsonization and the Mononuclear Phagocyte System (MPS) clearance.
- Troubleshooting: Incorporate "stealth" coatings like polyethylene glycol (PEG) (≥ 5 mol% density) to reduce protein adsorption. Ensure the PEG chain length (typically 2000-5000 Da) is sufficient to create a hydrophilic corona. Verify that your targeting ligand (e.g., peptide, antibody fragment) is not itself immunogenic or sticky to serum proteins.
Antibody-Drug Conjugates (ADCs)
Q3: My ADC shows in vitro potency but lacks in vivo efficacy in my heterogenous vasculature tumor model. A: This can stem from poor tumor penetration or linker instability.
- Troubleshooting:
- Assess Linker Stability: Run a plasma stability assay. Incubate the ADC in mouse/human plasma at 37°C, take time-point samples, and analyze by mass spectrometry or hydrophobic interaction chromatography for premature payload release.
- Evaluate Tumor Penetration: Use an ADC with a fluorescently labeled antibody component and perform immunohistochemistry on tumor sections to visualize distribution relative to heterogeneous blood vessels (CD31 stain).
Q4: I observe high toxicity in healthy tissues at doses lower than expected (narrow therapeutic window). A: This often indicates off-target toxicity due to antigen shedding, non-internalization in healthy tissues, or payload metabolism.
- Troubleshooting: Quantify soluble target antigen in the serum of your model. Consider switching to a more stable linker (e.g., enzyme-cleavable over pH-sensitive) or a less potent payload if antigen is widely expressed at low levels.
Viral Vectors (e.g., AAV, Lentivirus)
Q5: My systemic delivery of viral vectors for gene therapy results in low tumor transduction and high hepatic sequestration. A: Viral vectors, especially AAV, naturally tropism for the liver, and heterogenous tumor vasculature limits extravasation.
- Troubleshooting: Consider two strategies:
- Vector Engineering: Use AAV capsid serotypes with lower hepatic tropism (e.g., AAVrh.10) or perform directed evolution to select for capsids that bind tumor endothelial markers.
- Administration Route: For localized tumors, investigate intratumoral injection. For systemic delivery, pre-administer vasodilators (e.g., Nitroglycerin) or use tumor vascular disrupting agents (VDAs) in a metronomic schedule to potentially enhance extravasation.
Q6: I encounter low viral titer during production. A: This bottleneck is common in lentivirus or adenovirus production.
- Troubleshooting Protocol - Lentivirus Concentration:
- Harvest: Collect supernatant from producer cells (e.g., HEK293T) 48-72h post-transfection.
- Clarify: Centrifuge at 500 x g for 10 min to remove cell debris. Filter through a 0.45 µm PES filter.
- Concentrate: Use ultracentrifugation (70,000 x g for 2h at 4°C over a 20% sucrose cushion) or tangential flow filtration. Resuspend the pellet in cold PBS or medium.
- Titer: Determine functional titer via flow cytometry (for a fluorescent reporter) or qPCR for viral genomes.
Quantitative Data Comparison
Table 1: Platform Characteristics for Heterogenous Tumor Targeting
| Feature | Nanoparticles | Antibody-Drug Conjugates (ADCs) | Viral Vectors (AAV) |
|---|---|---|---|
| Typical Size Range | 10 - 200 nm | 10 - 15 nm (≈ Antibody size) | 20 - 25 nm (capsid) |
| Drug Payload Capacity | High (10,000s molecules) | Low (2-4 molecules per Ab) | N/A (Genetic payload) |
| Key Targeting Mechanism | Passive (EPR) & Active (surface ligands) | Active (Antigen-Antibody binding) | Active (Capsid-receptor binding) |
| Primary Limitation for Heterogeneous Vasculature | Inconsistent EPR effect; MPS clearance | Limited tumor penetration (~100 µm) | Neutralizing antibodies; Off-target transduction |
| Ideal Tumor Vasculature Profile | Leaky vasculature (high permeability) | Uniform, high antigen expression on endothelial/tumor cells | Accessible vasculature with specific receptors |
| Representative Clinical Examples | Doxil (liposome), Abraxane (albumin NP) | Enhertu (T-DXd), Adcetris (BV) | Luxturna (retinal gene therapy), Zolgensma (systemic) |
Experimental Protocols
Protocol 1: Assessing Tumor Vasculature Permeability to Nanoparticles
Objective: To quantify the Enhanced Permeation and Retention (EPR) effect in a xenograft model with heterogeneous vasculature.
- Inject: Administer fluorescently labeled, inert nanoparticles (e.g., 100 nm DiD-labeled PEGylated liposomes) intravenously.
- Image: At multiple time points (1, 4, 24, 48h), perform in vivo fluorescence imaging.
- Sacrifice & Section: At terminal time point, harvest tumors and snap-freeze for cryosectioning.
- Co-stain: Stain tumor sections for blood vessels (anti-CD31 antibody) and nuclei (DAPI).
- Quantify: Use confocal microscopy and image analysis software (e.g., ImageJ) to calculate:
- Extravasation Ratio: Fluorescence intensity outside CD31+ areas vs. inside.
- Penetration Depth: Distance of nanoparticle signal from nearest vessel wall.
Protocol 2: In Vivo Efficacy Study for an ADC in a Model with Heterogenous Antigen Expression
Objective: To evaluate ADC efficacy and correlate with target antigen distribution.
- Model Establishment: Implant tumor cells with known heterogeneous target antigen expression.
- Randomization: When tumors reach ~200 mm³, randomize animals into groups (n≥5): Vehicle, Isotype-Control ADC, Therapeutic ADC.
- Dosing: Administer treatments via intravenous injection at determined dose levels (e.g., 5-10 mg/kg ADC) weekly.
- Monitor: Measure tumor volumes and body weights 2-3 times weekly.
- Endpoint Analysis: At study end, process tumors for IHC. Serial sections should be stained for:
- Target Antigen
- Blood Vessels (CD31)
- Apoptosis (Cleaved Caspase-3)
- Proliferation (Ki-67)
- Correlative Analysis: Overlay antigen density maps (from IHC) with apoptosis signals to determine if efficacy correlates with antigen-positive regions.
Visualizations
Diagram 1: Tumor Targeting Pathways
Title: Drug Delivery Platform Mechanisms
Diagram 2: ADC Efficacy Workflow
Title: ADC Mechanism of Action from Injection to Killing
The Scientist's Toolkit: Key Reagent Solutions
| Reagent/Material | Primary Function in Context of Tumor Vasculature Research |
|---|---|
| PEGylated Phospholipids (e.g., DSPE-PEG2000) | Core component for creating "stealth" nanoparticles that evade immune clearance, critical for circulating long enough to reach heterogenous tumor sites. |
| pH-Sensitive Linkers (e.g., Valine-Citruline) | Used in ADCs and some NPs to ensure stable circulation but selective drug release in the acidic tumor microenvironment or lysosomal compartment. |
| Anti-CD31 Antibody | Standard immunohistochemistry marker for pan-endothelial cells, essential for quantifying tumor vascular density, morphology, and normalization. |
| Recombinant AAV Serotype Library | Enables screening for capsids with enhanced tropism for specific tumor vascular endothelial markers, overcoming natural transduction biases. |
| Fluorescent Lipophilic Tracers (e.g., DiD, DiR) | Labels nanoparticles for in vivo and ex vivo tracking of biodistribution, tumor accumulation, and penetration depth via imaging. |
| Matrigel Basement Membrane Matrix | Used in vitro to create 3D endothelial tubulogenesis assays to study the effect of drugs on vessel formation and permeability. |
| Vascular Disrupting Agent (e.g., CA4P) | Tool compound to acutely modulate tumor vasculature permeability, used experimentally to test if "priming" improves delivery platform uptake. |
Technical Support Center: Troubleshooting Guides & FAQs
Q1: In dynamic contrast-enhanced MRI (DCE-MRI) for assessing tumor vasculature, we observe high spatial heterogeneity in the Ktrans (volume transfer constant) map. How do we determine if this is a true biological signal or an artifact of motion or poor contrast agent bolus?
- A: True heterogeneity is a key focus in compensating for tumor vasculature heterogeneity. First, verify the contrast agent injection protocol: use a power injector at 3-5 mL/s followed by a saline flush for a consistent bolus. Check for patient motion artifacts by reviewing pre-contrast T1-weighted sequences; implement image registration if motion is detected. Analytically, plot the arterial input function (AIF); a poor, dispersed bolus will show a low, broad peak. For validation, correlate a sub-region of high Ktrans with a separate modality (e.g., a region of high vessel density on co-registered diffusion-weighted MRI or from a biopsy sample). Persistent, structured spatial patterns (e.g., rim-high, central-low) are likely biological.
Q2: When isolating circulating tumor cells (CTCs) for vascular heterogeneity studies, our CellSearch or microfluidic chip yield is unexpectedly low. What are the primary troubleshooting steps?
- A: Low CTC yield compromises analysis of intravasation, a critical process in vasculature research.
- Pre-analytical Variables: Ensure blood collection tubes (CellSave or other EDTA tubes) are inverted 8-10 times immediately and processed within strict time windows (96h for CellSave, <4h for EDTA). Do not chill samples.
- Sample Volume: For metastatic cancers, 7.5 mL is standard, but for early-stage or assessing heterogeneity, consider processing 30 mL across multiple tubes.
- EpCAM Expression: Remember that epithelial-to-mesenchymal transition (EMT), driven by heterogeneous microenvironments, can downregulate EpCAM. Integrate a size-based or label-free enrichment method in parallel to capture EpCAM-low CTC populations relevant to heterogeneous vasculature.
- Downstream Analysis: If using staining, optimize antibody concentrations for your target (CK, CD45) and include a nuclear stain (DAPI) to confirm cell integrity.
Q3: Our measurements of circulating endothelial cells (CECs) and circulating endothelial progenitor cells (CEPCs) by flow cytometry show high variability between replicates. How can we standardize this?
- A: CECs/CEPCs are direct biomarkers of vascular injury and repair. Variability often stems from pre-processing and gating.
- Protocol: Use a standardized lyse-no-wash procedure to minimize cell loss. Process samples within 4 hours of draw.
- Antibody Panel: Use a consensus panel: CD45-, CD31+, CD146+, CD133- for mature CECs; CD45-, CD34+, CD133+, VEGFR2+ for CEPCs. Include 7-AAD for viability.
- Gating Strategy: First, gate on nucleated cells (7-AAD+), then remove CD45+ leukocytes. Use fluorescence minus one (FMO) controls to accurately set boundaries for positive populations, especially for dim markers like VEGFR2. Absolute counting with precision beads is mandatory.
Q4: For analyzing exosomes as carriers of angiogenic biomarkers, how do we differentiate tumor-derived exosomes from other bodily fluid exosomes?
- A: This is central to understanding vascular niche signaling.
- Isolation: Use sequential ultracentrifugation (see protocol below) or size-exclusion chromatography for purity.
- Characterization: Validate by nanoparticle tracking analysis (size mode: 80-150 nm) and transmission electron microscopy.
- Tumor-Specific Markers: Probe for tumor-associated proteins (e.g., EGFRvIII, PSMA) or tumor-specific miRNAs (e.g., miR-21, miR-210) known to be involved in angiogenic pathways. Immunocapture using antibodies against tumor-specific surface tetraspanins (e.g., CD151) can pre-enrich.
Detailed Experimental Protocols
Protocol 1: Sequential Ultracentrifugation for Plasma-Derived Exosome Isolation
- Collect blood in EDTA tubes, process within 30 min. Centrifuge at 2,000 x g for 20 min at 4°C to obtain platelet-poor plasma.
- Transfer plasma to a new tube. Centrifuge at 12,000 x g for 45 min at 4°C to remove cell debris and large apoptotic bodies.
- Carefully collect supernatant, filter through a 0.22 µm PES filter.
- Ultracentrifuge the filtrate at 110,000 x g for 70 min at 4°C (Type 70 Ti rotor or equivalent).
- Discard supernatant, resuspend pellet in large volume of PBS. Ultracentrifuge again at 110,000 x g for 70 min at 4°C.
- Discard supernatant, resuspend final exosome pellet in 50-100 µL PBS. Aliquot and store at -80°C.
Protocol 2: Immunohistochemistry (IHC) for Consecutive Staining of Vascular Markers
- Use formalin-fixed, paraffin-embedded (FFPE) tumor sections (4 µm) mounted on charged slides.
- Deparaffinize and rehydrate through xylene and graded ethanol series.
- Perform heat-induced epitope retrieval (HIER) in citrate buffer (pH 6.0) or EDTA (pH 9.0) for 20 min.
- Block endogenous peroxidase with 3% H2O2 for 10 min, then block with 5% normal serum for 30 min.
- Incubate with primary antibody (e.g., CD31 for endothelium) overnight at 4°C.
- Develop with HRP-polymer and DAB chromogen (brown signal).
- To strip antibodies for consecutive staining, place slide in HIER buffer and heat for 20 min.
- Cool, wash, and repeat steps 4-6 with a second primary antibody (e.g., α-SMA for pericytes) using a different chromogen (e.g., Vector Red).
- Counterstain with hematoxylin, dehydrate, and mount.
Quantitative Data Summary
Table 1: Comparison of Key Imaging Biomarkers for Tumor Vasculature
| Biomarker | Imaging Modality | Typical Parameter | Normal Range | Tumor Vasculature Indication | Key Challenge |
|---|---|---|---|---|---|
| Perfusion | DCE-MRI | Ktrans (min-1) | Low (organ-dependent) | High = leaky vasculature | AIF selection, motion |
| Blood Volume | DCE-MRI, DSC-MRI | vp (%) | ~5% | Elevated | Contrast agent extravasation |
| Hypoxia | PET (¹⁸F-FMISO) | Tumor-to-Muscle Ratio | ~1.0 | >1.2 = hypoxic | Slow clearance, background |
| Metabolism | PET (¹⁸F-FDG) | SUVmax | Variable | High = glycolytically active | Not vessel-specific |
Table 2: Performance of Circulating Biomarker Assays
| Biomarker | Sample Type | Common Assay | Typical Detection Limit | Analytical Turnaround | Primary Research Utility |
|---|---|---|---|---|---|
| CTCs | Whole Blood | CellSearch | 1 CTC / 7.5 mL | 24-48 h | Prognosis, heterogeneity |
| ctDNA | Plasma | ddPCR | 0.1% allele frequency | 1-3 days | Mutational tracking |
| Exosomes | Plasma/Serum | NTA + ELISA | 106 particles/mL | 2-4 days | Inter-cellular signaling |
| CECs | Whole Blood | Flow Cytometry | 10 cells / mL | <6 hours | Vascular injury monitor |
Pathway & Workflow Diagrams
Diagram Title: Biomarker Validation Workflow for Heterogeneity Research
Diagram Title: Hypoxia-Driven Angiogenic Signaling Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Biomarker Experiments in Vascular Research
| Item | Function / Application | Example Vendor/Product |
|---|---|---|
| EDTA or CellSave Blood Collection Tubes | Preserves cell integrity and prevents clotting for CTC and cfDNA analysis. | BD Vacutainer EDTA; Menarini CellSave Tubes |
| Recombinant Human VEGF | Positive control for endothelial cell tube formation assays and angiogenic signaling studies. | PeproTech |
| Anti-human CD31/PECAM-1 Antibody | Gold-standard marker for immunohistochemical staining of vascular endothelium. | Agilent Dako, Clone JC70A |
| Anti-human EpCAM Antibody (conjugated) | Magnetic or fluorescent capture/detection of epithelial-derived CTCs. | Miltenyi Biotec (microbeads); BioLegend (clone 9C4) |
| Cell-Free DNA Collection Tubes | Stabilizes nucleases to prevent genomic DNA contamination in plasma for ctDNA studies. | Streck cfDNA BCT |
| Exosome Isolation Reagent (PEG-based) | Simplified, column-free precipitation of exosomes from serum/plasma/culture media. | Thermo Fisher Total Exosome Isolation Kit |
| Matrigel Basement Membrane Matrix | 3D matrix for in vitro endothelial cell tube formation assays to model angiogenesis. | Corning Matrigel |
| Fluorophore-conjugated Annexin V | Detection of phosphatidylserine exposure on circulating endothelial cell (CEC) apoptosis. | BD Pharmingen |
Technical Support Center
FAQs & Troubleshooting Guide
Q1: In our colorectal cancer mouse model, anti-VEGF monotherapy initially reduces tumor volume, but resistance develops rapidly. What are the potential compensatory mechanisms and how can we investigate them? A1: A common compensatory mechanism is the upregulation of alternative pro-angiogenic pathways, such as Angiopoietin-2 (Ang2). To investigate:
- Assess Pathway Activation: Perform IHC/IF staining on treated vs. control tumor sections for markers like pERK (downstream of VEGF) and Tie2 receptor phosphorylation. A decrease in pERK with sustained or increased pTie2 suggests Ang2 compensation.
- Analyze Vessel Phenotype: Use immunofluorescence (CD31/α-SMA) to assess pericyte coverage. Anti-VEGF may prune immature vessels, leaving a normalized, pericyte-covered network initially, but resistance often leads to re-emergence of abnormal vasculature.
- Protocol - Multiplex IHC/IF:
- Tissue: FFPE tumor sections (5 µm).
- Antibodies: Primary antibodies: Rabbit anti-CD31 (vascular endothelial), Rat anti-α-SMA (pericytes), Goat anti-Ang2.
- Detection: Use species-specific secondary antibodies conjugated to distinct fluorophores (e.g., Alexa Fluor 488, 555, 647).
- Imaging & Quantification: Use a confocal microscope. Quantify Ang2 fluorescence intensity in the tumor stroma and perivascular area using image analysis software (e.g., ImageJ).
Q2: We are testing a dual anti-VEGF/anti-Ang2 agent in glioblastoma (GBM). Our perfusion data (using dextran-FITC) is inconsistent. What could be the issue? A2: Inconsistent perfusion data in GBM is often due to persistent vascular heterogeneity and high intracranial pressure. Follow this troubleshooting guide:
- Check Injection Consistency: Ensure the dextran-FITC (or similar perfusion marker like Hoechst 33342) is administered via a consistent route (tail vein) at a fixed volume and rate (e.g., 100 µL/10 sec).
- Control for Time: Sacrifice animals at a strict, uniform interval post-injection (e.g., 1 minute). Even slight variations can cause major differences due to the abnormal blood flow dynamics in tumors.
- Co-stain for Vessel Lumen: Include a primary antibody against a pan-endothelial marker (CD31) to distinguish perfused (FITC+ CD31+) versus non-perfused (FITC- CD31+) vessels.
- Protocol - Perfusion Assessment:
- Inject 100 µL of 50 mg/mL FITC-labeled dextran (70 kDa) via tail vein.
- After 60 seconds, euthanize the mouse and immediately harvest the tumor.
- Snap-freeze in O.C.T. compound. Section (10 µm).
- Fix in ice-cold acetone for 10 min, block, and stain with anti-CD31 antibody.
- Image multiple, random fields from the tumor core and periphery.
Q3: When analyzing tumor hypoxia after vascular normalization therapy, our pimonidazole staining shows unexpected patterns. What controls and quantification methods are critical? A3: Pimonidazole staining requires careful controls. Unexpected patterns (e.g., increased hypoxia after therapy) can be real or artifactual.
- Essential Controls:
- Negative Control: Include a tumor from a non-pimonidazole-injected mouse to check for non-specific antibody binding.
- Positive Control: Use a known hypoxic tumor (e.g., untreated, large tumor) to ensure the staining protocol works.
- Critical Quantification Method:
- Do not rely on "% positive area" alone. This can be misleading if therapy changes vessel distribution.
- Measure Hypoxic Radius: Calculate the distance from each perfused vessel (CD31+) to the nearest pimonidazole+ region. A successful normalizing agent should reduce the median hypoxic radius.
- Protocol - Hypoxic Radius Measurement:
- Perform dual IF for CD31 and pimonidazole adducts.
- In image analysis software (e.g., QuPath, ImageJ with plugins), threshold the CD31 signal to create vessel masks.
- Use a distance transform function to create a map of distances from every pixel to the nearest vessel.
- Overlay the pimonidazole-positive mask and extract the distance values for all hypoxic pixels. Plot the frequency distribution.
Table 1: Preclinical Efficacy of Vascular Normalizing Agents
| Tumor Type (Mouse Model) | Agent Class | Key Metric Change (vs. Control) | Proposed Mechanism of Action |
|---|---|---|---|
| Colorectal Carcinoma (MC38) | Anti-VEGF (Bevacizumab) | - Tumor Growth: ~40% inhibition; - Vessel Density: -50%; - Pericyte Coverage: +25% | Prunes immature vessels, stabilizes remaining vasculature. |
| Glioblastoma (GL261) | Anti-Ang2 (REGN910) | - Tumor Growth: ~30% inhibition; - Vessel Leakiness: -60%; - T-cell Infiltration: +3-fold | Blocks Tie2 signaling, reduces endothelial inflammation, improves vessel integrity. |
| Breast Carcinoma (4T1) | Anti-VEGF/Anti-Ang2 Bispecific | - Tumor Growth: ~60% inhibition; - Lung Metastasis: -80%; - Median Survival: +150% | Dual inhibition prevents compensatory signaling, enhances normalization window, improves chemotherapy delivery. |
| Renal Cell Carcinoma (RENCA) | VEGF Receptor TKI (Sunitinib) | - Tumor Growth: ~55% inhibition; - Hypoxic Area: Initial increase, then decrease | Rapid vessel pruning causes transient hypoxia, followed by re-normalization. |
Table 2: Clinical Trial Biomarkers of Response
| Agent | Tumor Type (Trial Phase) | Correlative Biomarker of Positive Response | Association with Outcome |
|---|---|---|---|
| Bevacizumab (anti-VEGF) + Chemo | Metastatic Colorectal Cancer (Phase III) | High baseline plasma VEGF-A; Reduction in circulating endothelial cells (CECs) after 1 cycle | Associated with longer progression-free survival (PFS). |
| Faricimab (anti-VEGF/Ang2) | Glioblastoma (Phase II) | Reduction in dynamic contrast-enhanced MRI (DCE-MRI) parameter Ktrans (vascular permeability) | Greater reduction in Ktrans correlated with improved overall survival (OS) trend. |
| Nesvacumab (anti-Ang2) + Aflibercept (VEGF trap) | Ovarian Cancer (Phase I) | Decrease in plasma Ang2 and VEGF-D levels post-treatment | Biomarker decrease correlated with stable disease. |
The Scientist's Toolkit: Key Research Reagent Solutions
| Item & Common Example | Function in Vascular Normalization Research |
|---|---|
| Dextran, Fluorescein-labeled (e.g., 70 kDa FITC-Dextran) | A perfusion tracer. Injected intravenously to visualize functional, patent blood vessels. High molecular weight limits extravasation. |
| Pimonidazole Hydrochloride | A hypoxia probe. Forms protein adducts in cells with pO₂ < 10 mm Hg. Detected by specific antibodies to map hypoxic regions. |
| α-Smooth Muscle Actin (α-SMA) Antibody | Marks pericytes and vascular smooth muscle cells via immunofluorescence. Critical for quantifying vessel maturity and normalization. |
| CD31/PECAM-1 Antibody | Pan-endothelial cell marker. Used to visualize total tumor vasculature, regardless of perfusion status. |
| Phospho-specific Antibodies (e.g., pVEGFR2, pTie2) | Detect activation states of key receptors in VEGF and Ang2 signaling pathways. Assesses target engagement and compensatory signaling. |
| Recombinant VEGF / Ang2 Protein | Used as positive controls in Western blot or ELISA, or to stimulate cells in vitro to validate antibody/inhibitor functionality. |
| Matrigel | Used for in vitro endothelial tube formation assays to assess the functional impact of agents on angiogenesis. |
Experimental Protocols
Protocol 1: Multiparameter Immunohistochemistry Analysis of Vessel Normalization Objective: To simultaneously quantify vessel density, pericyte coverage, and hypoxia in a single tumor section. Steps:
- Tumor Processing: Harvest tumor, fix in 4% PFA for 24h, paraffin-embed. Section at 5 µm.
- Deparaffinization & Antigen Retrieval: Use standard xylene/ethanol series. Perform heat-induced epitope retrieval in citrate buffer (pH 6.0).
- Sequential Staining (Multiplex IF): a. Round 1: Block with 3% BSA, incubate with primary antibody (e.g., Rabbit anti-CD31), then with HRP-conjugated secondary. Detect with Tyramide signal amplification (TSA) using Fluorophore A (e.g., Cy3). Heat-inactivate the HRP (10 min, 95°C in retrieval buffer). b. Round 2: Block again. Incubate with second primary (e.g., Rat anti-α-SMA), then secondary antibody conjugated to a different fluorophore (e.g., Alexa Fluor 488). c. Round 3 (Hypoxia): Repeat process with antibody against pimonidazole adducts (if animal was injected with pimonidazole prior to sacrifice) using Fluorophore C (e.g., Cy5).
- Imaging & Analysis: Use a fluorescent slide scanner. Use spectral unmixing if necessary. Quantify: CD31+ area (vessel density), α-SMA+ area adjacent to CD31+ (pericyte coverage), and pimonidazole+ area.
Protocol 2: Dynamic Contrast-Enhanced MRI (DCE-MRI) in Preclinical Models Objective: To non-invasively assess tumor vascular permeability (Ktrans) and blood volume in response to therapy. Steps:
- Animal Preparation: Anesthetize mouse and place in MRI-compatible holder with temperature control.
- Baseline Scanning: Acquire T1-weighted images to establish baseline tissue signal.
- Contrast Agent Injection: Administer a bolus of gadolinium-based contrast agent (e.g., Gd-DTPA, 0.1 mmol/kg) via a tail vein catheter during scanning.
- Image Acquisition: Rapidly acquire repeated T1-weighted images over 20-30 minutes to track contrast agent kinetics in the tumor and a reference artery (e.g., vena cava).
- Data Analysis: Use pharmacokinetic modeling (e.g., Tofts model) on signal intensity curves to calculate the transfer constant Ktrans (min⁻¹), a measure of vascular permeability and blood flow.
Pathway and Workflow Diagrams
Title: VEGF and Ang2 Signaling Pathways in Endothelial Cells
Title: Workflow for Assessing Vascular Normalization In Vivo
Technical Support Center: Troubleshooting & FAQs
This support center addresses common experimental challenges in validating vascular targets within the context of Compensating for Tumor Vasculature Heterogeneity research.
FAQ & Troubleshooting Guide
Q1: In our 3D co-culture assay, we observe inconsistent vessel network formation when testing DLL4 inhibitors. What could be the cause? A: Inconsistent network formation often stems from variable endothelial cell (EC) to pericyte ratios or off-target effects. Ensure:
- Use a defined EC:pericyte ratio (e.g., 4:1) and confirm cell viability >95% before seeding.
- Validate DLL4 inhibitor specificity. Use a VEGF-A trap (e.g., Aflibercept) as a control; DLL4 inhibition should produce a characteristically dense, hyper-sprouting phenotype distinct from VEGF inhibition.
- Quantify networks using standardized metrics (see Table 1).
Q2: Our flow cytometry analysis of Tumor-Endothelial Cells (TEMs) shows high background noise. How can we improve purity and detection? A: High background is common due to non-specific antibody binding and dead cells.
- Protocol Enhancement: Include a live/dead cell viability dye (e.g., Zombie NIR) before surface antibody staining. Use Fc receptor blocking (e.g., Human TruStain FcX).
- Gating Strategy: Use a sequential gating strategy: Singlets → Live cells → CD45- (non-hematopoietic) → CD31+ (pan-endothelial) → TEM markers (e.g., CD109+ or high CD276+). Always include Fluorescence Minus One (FMO) controls for each marker.
Q3: When attempting to modulate pericyte coverage in our murine xenograft model, we see no change in drug delivery efficiency despite successful PDGFR-β inhibition. Why? A: This highlights vasculature heterogeneity. Pericyte modulation can lead to vascular normalization or regression.
- Troubleshoot: Measure multiple parameters. Successful PDGFR-β inhibition may reduce coverage, but if it induces hypoxia and vessel collapse, perfusion may not improve.
- Required Analysis: Correlate pericyte coverage (α-SMA+ area/CD31+ area) with:
- Perfusion: Lectin (e.g., Lycopersicon esculentum) injection prior to sacrifice.
- Hypoxia: Pimonidazole staining.
- Outcome: See Table 2. Improved delivery is only expected in a "normalization window."
Q4: Our Western blot for cleaved Notch1 Intracellular Domain (NICD) in treated endothelial cells shows weak or no signal. A: NICD is nuclear and transient. Optimize protocol:
- Sample Preparation: Use nuclear extraction kits. Include a positive control (e.g., cells treated with a known γ-secretase activator or recombinant DLL4).
- Timing: NICD peaks 30-120 minutes post-DLL4 stimulation. Perform a time-course experiment.
- Antibodies: Validate antibodies on cell lines with siRNA-mediated NOTCH1 knockdown.
Experimental Protocols
Protocol 1: 3D Fibrin Gel Bead Sprouting Assay for DLL4-Notch Inhibition
- Coat cytodex beads with human umbilical vein endothelial cells (HUVECs). Culture until confluent.
- Embed beads in a fibrinogen/thrombin gel in 24-well plates. Include pericytes (e.g., human placental pericytes) in the gel at a 4:1 (EC:Pericyte) ratio.
- After polymerization, add EGM-2 medium supplemented with VEGF (50 ng/mL) and FGF-2 (30 ng/mL).
- Add DLL4 blocking antibody (e.g., Demcizumab) or Isotype control. Use a VEGF inhibitor (e.g., Bevacizumab) as a phenotypic control.
- Culture for 5-7 days, refreshing medium and treatments every 48 hours.
- Fix with 4% PFA. Stain for CD31 (EC marker) and α-SMA (pericyte marker).
- Image with confocal microscopy. Quantify: total sprout length per bead, number of sprouts, and pericyte association index.
Protocol 2: Isolation and Characterization of TEMs from Tumor Digests
- Generate single-cell suspension from human or murine tumor using a gentleMACS Dissociator with enzymatic cocktail (Collagenase IV/DNase I).
- Perform density gradient centrifugation (e.g., Percoll) to enrich for viable cells.
- Resuspend in FACS buffer (PBS + 2% FBS). Incubate with viability dye for 15 min at 4°C.
- Wash, then incubate with Fc block for 10 min.
- Stain with conjugated antibodies: CD45-APC/Cy7, CD31-PE/Cy7, CD109-BV421 (human) or CD45-APC/Cy7, CD31-PE/Cy7, CD276-BV421 (murine). Incubate 30 min at 4°C in the dark.
- Wash twice, resuspend, and filter through a 35µm cell strainer.
- Sort or analyze on a flow cytometer. Gate as per FAQ Q2.
Data Presentation
Table 1: Quantitative Phenotypes of Vasculature Following Targeted Inhibition
| Target | Agent Example | Sprout Density (% vs Control) | Vessel Diameter (µm) | Pericyte Coverage Index | Functional Outcome (Perfusion) |
|---|---|---|---|---|---|
| VEGF | Bevacizumab | -65% | 8.2 ± 1.5 | 0.85 ± 0.10 | Severely Reduced |
| DLL4 | Demcizumab | +220% | 5.1 ± 0.8 | 0.55 ± 0.12 | Increased, But Chaotic |
| PDGFR-β | CP-673451 | -40% | 12.5 ± 2.3 | 0.35 ± 0.08 | Variable (Context-Dependent) |
Table 2: Correlating Pericyte Modulation with Drug Delivery Outcomes
| Pericyte Coverage Change | Hypoxia Status | Vessel Maturation Score | Doxorubicin Penetration (vs Baseline) | Recommended Next Step |
|---|---|---|---|---|
| Severe Decrease (>60%) | Increased (pimo+) | Low | -50% | Co-administer vessel-stabilizing agent (e.g., Ang-1). |
| Moderate Decrease (30-50%) | Unchanged or Reduced | Medium | +100% | Optimal "Normalization Window" for therapy. |
| No Change | Unchanged | High | No Change | Increase dose or switch to anti-angiogenic. |
Visualizations
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function/Application | Example Product/Catalog # |
|---|---|---|
| Recombinant Human DLL4 (Fc chimera) | Acts as a soluble Notch activator/ligand for control experiments in sprouting assays. | R&D Systems, 1506-D4-050 |
| Anti-Human DLL4 Neutralizing Antibody | Specifically blocks DLL4-Notch1 interaction to induce hyper-sprouting phenotype. | Bio-Techne, MAB1388 (Demcizumab analog) |
| Lectin from Lycopersicon esculentum (TRITC) | Labels perfused blood vessels when injected intravenously prior to sacrifice. | Vector Laboratories, FL-1171 |
| Anti-Mouse/Rat CD276 (B7-H3) Antibody | Key for identifying murine TEM populations via flow cytometry. | BioLegend, 124207 |
| Recombinant PDGF-BB & PDGFR-β Inhibitor | For modulating pericyte recruitment and dissociation in vitro. | PeproTech, 100-14B (PDGF-BB); MedChemExpress, CP-673451 (Inhibitor) |
| Pimonidazole HCl | Hypoxia probe. Forms adducts in cells with pO₂ < 10 mm Hg, detectable by antibody. | Hypoxyprobe, HP3-100Kit |
| 3D Fibrinogen from Human Plasma | Matrix for 3D in vitro sprouting assays, providing a physiological environment. | Sigma-Aldrich, F3879 |
| Zombie NIR Fixable Viability Kit | Distinguishes live/dead cells for flow cytometry, reducing background from dead cells. | BioLegend, 423105 |
Clinical Trial Design Considerations for Heterogeneity-Addressing Therapies
Troubleshooting Guides & FAQs
Q1: During patient stratification for our vascular-targeting therapy trial, biomarker expression is inconsistent across tumor biopsy samples. How should we define eligibility?
A: This is a common issue due to intratumoral heterogeneity. Standard practice is to define a positivity threshold (e.g., ≥50% of sampled tumor area shows target biomarker expression via IHC). For circulating biomarkers, use the mean of three baseline measurements. Consider adaptive designs that allow eligibility criteria refinement.
Q2: Our primary endpoint (PFS) shows high variance in the treatment arm, likely due to heterogeneous response. What alternative statistical models can we use?
A: High variance suggests differential subpopulation response. Consider mixture cure models or time-varying covariate models in your analysis plan. Using a co-primary endpoint (e.g., PFS + volumetric perfusion MRI change at 8 weeks) can better capture biologic effect.
Q3: How do we handle dose selection when the therapy's effect is modulated by variable tumor vasculature density?
A: Implement a Phase Ib "basket" design with pharmacodynamic (PD) escalation. Dose is escalated within predefined vascular density cohorts (e.g., low, medium, high angiogenic score). The MTD/RP2D is determined per cohort.
Q4: What is the recommended control arm for a trial testing an anti-heterogeneity agent combined with a standard therapy?
A: The control should be standard therapy + placebo. A 2x2 factorial design (e.g., [Standard ± Novel Agent]) can efficiently test the additive effect. Stratified randomization by key heterogeneity factors (e.g., baseline perfusion status, genomic subtype) is critical.
Q5: Imaging-based PD biomarkers (e.g., DCE-MRI) have high intra-patient variability. How do we set a reliable threshold for biological activity?
A: Establish a quantitative, centralized imaging core lab. The threshold for biological activity should be based on the coefficient of variation (CV) from test-retest studies. A change > 2x the CV (typically 10-15% for Ktrans) is considered reliable.
Table 1: Common Biomarkers for Tumor Vasculature Heterogeneity & Trial Applications
| Biomarker | Assay Method | Typical Threshold for Positivity | Role in Trial Design | Associated Challenge |
|---|---|---|---|---|
| Microvessel Density (MVD) | CD31/IHC | ≥20 vessels/HPF (high) | Stratification factor | Intratumoral spatial heterogeneity |
| VEGFA Expression | RNA-seq / ISH | Top 40% of expression in cohort | Enrichment biomarker | Discordance between primary/met |
| Perfusion (Ktrans) | DCE-MRI | ≥ 0.15 min⁻¹ (high) | Pharmacodynamic endpoint | Technical variability between scanners |
| Circulating Endothelial Cells (CECs) | Flow Cytometry | >50 cells/mL | Early efficacy signal | Pre-analytical variability |
Table 2: Comparison of Trial Designs for Heterogeneity-Addressing Therapies
| Design Type | Key Feature | Best Suited For | Sample Size Implication | Key Analytical Method |
|---|---|---|---|---|
| Stratified Enrichment | Pre-screening & randomization within biomarker strata | Biomarker defines a distinct subpopulation | Reduced, targets responsive group | Stratified Cox regression |
| Adaptive Biomarker | Interim analysis to modify biomarker thresholds/strata | Preliminary signal but unclear cutoff | Similar to traditional, requires alpha spending | Bayesian adaptive design |
| Basket (Cohort) | Single therapy tested in multiple biomarker-defined cohorts | Therapy targeting a common heterogeneous mechanism | Per cohort: 15-30 patients | Bayesian hierarchical modeling |
| Factorial | Tests combination & interaction of two+ interventions | Agent designed to overcome vascular heterogeneity-driven resistance | Larger, powered for interaction term | Logistic regression with interaction term |
Experimental Protocols
Protocol 1: Multiplex Immunofluorescence (mIF) for Spatial Heterogeneity Analysis of Tumor Vasculature Purpose: To quantitatively assess co-expression of vascular targets (e.g., CD31, αvβ3, CA9) and the tumor microenvironment in a single tissue section for patient stratification. Materials: Formalin-fixed, paraffin-embedded (FFPE) tumor sections, multiplex IHC/IF antibody panel, fluorescence microscope with spectral imaging. Steps:
- Deparaffinize and rehydrate FFPE sections.
- Perform antigen retrieval using pH 6.0 citrate buffer.
- Apply first primary antibody, incubate, then detect with tyramide signal amplification (TSA)-conjugated fluorophore (e.g., Cy5).
- Denature antibody complex via microwave treatment in retrieval buffer.
- Repeat steps 3-4 for each marker in the panel (Cy5, Cy3, FITC, etc.).
- Counterstain with DAPI, mount.
- Acquire whole-slide images using a spectral imager.
- Use image analysis software (e.g., HALO, QuPath) for spatial analysis: calculate MVD, biomarker co-expression within 10μm of vessels, and heterogeneity indices (e.g., Shannon Index).
Protocol 2: Dynamic Contrast-Enhanced MRI (DCE-MRI) for Pharmacodynamic Assessment Purpose: To measure changes in tumor vascular permeability/perfusion (Ktrans) as a PD biomarker of vascular-targeting therapy. Materials: 3T MRI scanner, gadolinium-based contrast agent, pharmacokinetic modeling software. Steps:
- Pre-baseline: Acquire T1 mapping sequences (variable flip angles) for baseline tissue T1 calculation.
- Dynamic Series: Administer contrast agent as a bolus (0.1 mmol/kg). Simultaneously, acquire fast T1-weighted gradient-echo sequences (e.g., TWIST, VIBE) every 5-10 seconds for 10 minutes.
- Arterial Input Function (AIF): Define a region of interest (ROI) in a major artery (e.g., femoral) from the dynamic images.
- Analysis: Use Tofts or extended Tofts model in software (e.g., Olea Sphere, MITK). Coregister all images. Draw ROI around entire tumor on each slice. Model computes pixel-by-pixel Ktrans, reflecting perfusion/permeability.
- PD Endpoint: Calculate the % change in median tumor Ktrans from baseline to Day 28. A decrease >30% is often considered a significant biological effect.
Diagrams
Title: Stratified Enrollment & Randomization Workflow
Title: Heterogeneity Drivers & Trial Design Implications
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Vasculature Heterogeneity Research | Example/Provider |
|---|---|---|
| Multiplex IHC/IF Panels | Simultaneous spatial profiling of vascular (CD31), pericyte (α-SMA), hypoxia (CA9), and immune (CD8) markers. | Akoya Biosciences (Phenocycler), Standardized from commercial vendors (Abcam, CST). |
| Matrigel (Growth Factor Reduced) | In vitro assay for endothelial tube formation to test anti-angiogenic compound efficacy. | Corning, BD Biosciences. |
| Anti-Human VEGFA Neutralizing Antibody | Positive control for in vitro and in vivo angiogenesis inhibition studies. | Bevacizumab (commercial), R&D Systems antibodies. |
| MRI Contrast Agents (Macromolecular) | For improved quantification of vascular permeability (Ktrans, ve) in DCE-MRI PD studies. | Gadofosveset (Ablavar), or preclinical agents like Ferumoxtran-10. |
| Circulating Endothelial Cell (CEC) Enrichment Kit | Isolate and quantify CECs from patient blood as a potential surrogate biomarker. | CD146-based immunomagnetic kits (e.g., from Miltenyi Biotec). |
| Next-Generation Sequencing Panels | Profile tumor for mutations in angiogenesis pathways (VHL, PBRM1) for cohort stratification. | Targeted panels (e.g., Illumina TST170, FoundationOne). |
| Pharmacokinetic Modeling Software | Analyze DCE-MRI or other dynamic imaging data to derive quantitative vascular parameters. | Olea Sphere, PMI, MITK. |
| Patient-Derived Xenograft (PDX) Models | Preclinical models retaining the original tumor's vascular heterogeneity for therapy testing. | Providers: The Jackson Laboratory, Champions Oncology. |
Conclusion
Compensating for tumor vasculature heterogeneity is not a single-strategy endeavor but requires a multifaceted, context-dependent approach. The foundational understanding of its dynamic biology must inform the selection and application of methodological tools—from vascular normalization to smart nanocarriers. Success hinges on rigorous experimental optimization and validation using clinically relevant models and biomarkers. The future lies in personalized combination strategies that are temporally controlled and guided by advanced imaging, moving beyond a one-size-fits-all model. By systematically addressing this heterogeneity, the field can significantly enhance the delivery and efficacy of next-generation oncology therapeutics, turning a major biological barrier into a tractable therapeutic target.